Nepal Gray Langur, Semnopithecus schistaceus
NEPAL GRAY LANGUR
Semnopithecus schistaceus

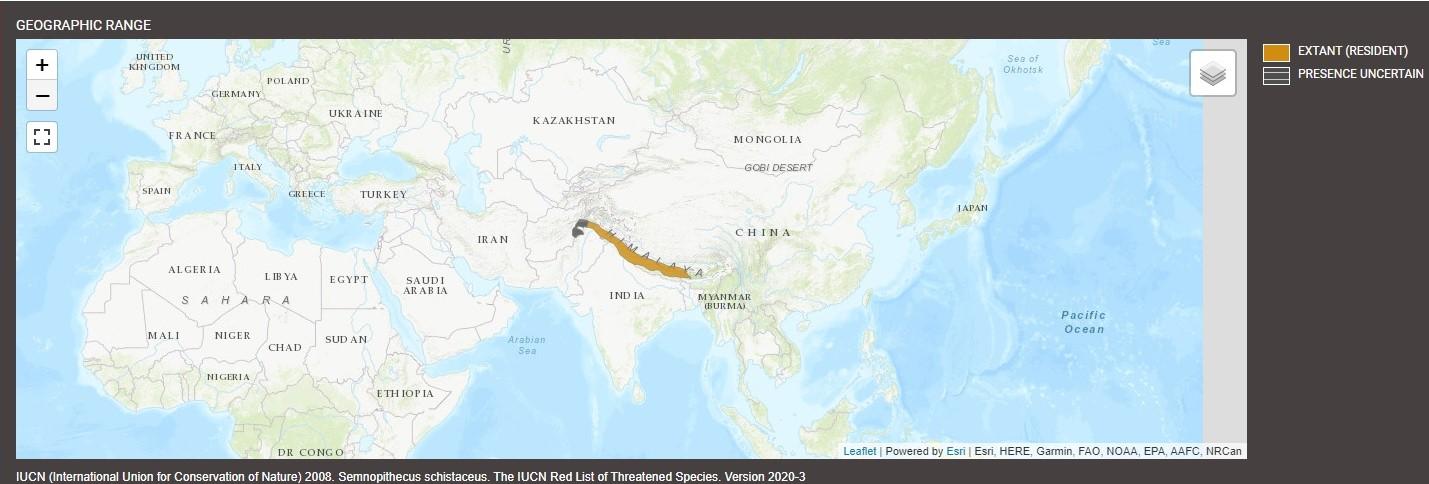
Geographic Distribution and Habitat
The Nepal gray langur (Semnopithecus schistaceus), also known as Himalayan gray langurs or central Himalayan langurs, is a species of langur native to the Himalayan region. Their range is quite extensive and includes Nepal, far southwestern Tibet, northern India, northern Pakistan, and Bhutan, and it may also extend into parts of northeastern Afghanistan, although their presence in this region is poorly documented. Current research suggests that the distribution of the Nepal gray langurs may not extend into Pakistan, but larger-scale surveys are needed to confirm their absence from the region. On the east side, their territory extends in India as far as the Buxa Tiger Reserve in northern West Bengal, reaching at least up to the Raidak River.
The Nepal gray langur thrives in these mountainous regions. They enjoy the forested areas in these regions, shrublands, and rocky areas (for example, inland cliffs or mountain peaks). They can be found at heights of 4,900 feet (1,500 m) and up to 13,000 feet (4,000 m). They prefer living in evergreen and broadleaf forests; however, they’ve been spotted in urban areas as well.
The Nepal gray langur was originally considered a subspecies of northern plain gray langurs, Semnopithecus entellus. More recent DNA studies, however, have demonstrated their independent status. There are currently no recognized subspecies.
Size, Weight, and Lifespan
Nepal gray langurs are among the largest in their genus. There are a few differences between males and females when it comes to body size and weight. Males have a head-to-body size of about 25 inches (69 cm), while females are usually a little bit smaller. Adult males weigh on average 39 pounds (18 kg), whereas females weigh 35 pounds (16kg). The heaviest langur ever recorded was a male Nepal gray langur, who weighed 58 pounds (26.5 kg). They have relatively long lifespans, at least in captivity, as they live up to 30 years.
Appearance
Nepal gray langurs owe their name to the characteristic dark gray color that covers their back and limbs. Their fur appears smooth and silky, and becomes creamy-white on their belly with a few brown undertones. Their faces, hands, and feet are bare; the exposed skin is pitch-black. There are no significant differences between the two sexes besides the slightly smaller size of females.
Nepal gray langurs have long tails that they use primarily for balance and to navigate their environment. Interestingly, northern populations of gray langurs, including the Nepal gray langur, carry their tails pointed forward, while southern populations tend to point them downward. The reason for this difference is not entirely clear; however, some scientists suggest it may be a cultural behavior adopted by different populations and passed down to their offspring.

Diet
This species is predominantly herbivorous, with a marked preference for leaves, including evergreen needles. Their diet is quite varied and includes fruits and fruit buds, leaf stalks, shoots and roots, seeds, acorns, grass, bamboo, fern rhizomes, mosses, and lichens. They also feed on invertebrate animals.
Leaves are their primary food source, but they can be rather stodgy. Leaves are hard to digest and contain various neurotoxins that can be quite taxing on mammals’ bodies. Langur species — including the Nepal gray langur — have the perfect evolutionary advantage: multi-chambered stomachs that allow bacteria to ferment food. In-stomach fermentation renders otherwise toxic leaves perfectly nutritious. This comes at a price, however, as digesting leaves makes these primates less active compared to other animals, since they do not provide ready-to-use energy. Troops that live near urban settlements also rely on cultivated plants.
They hardly ever drink water, relying instead on their moisture-rich diet for hydration. When they do, they might have a sip of water collected in tree cavities.
Behavior and Lifestyle
This species is diurnal and both terrestrial and arboreal, meaning that they are active during the day and spend their life in trees and on the ground. They are, however, not particularly active, as the majority of their time is spent resting due to their leaf-rich diet.
Their activity is influenced by the availability of food sources; for example, scarcity of food forces them to move for longer periods of time and pushes them further away. Conversely, troops that live near human settlements learn to rely more on cultivated plants, so they forage more in the same areas compared to those that feed exclusively on wild plants. Temperatures also have an impact on their routine: the higher the temperature, the more active they become.
Langurs spend much of their downtime grooming themselves and one another. Allogrooming (grooming each other) is a literal case of “you scratch my back, I scratch yours.” Grooming serves multiple functions: the recipient has dirt and parasites removed from their fur, while the groomer may gain an opportunistic snack by consuming those pests. Beyond hygiene, the physical contact and time invested help strengthen social bonds, particularly among close family members. In many ways, this mirrors how humans spend time with family, which helps maintain and reinforce social relationships.
Nepal gray langurs engage in geophagia (the consumption of soil or rocks) to obtain essential micronutrients. This behavior likely helps supplement the diet with nutrients such as sodium, which is vital for physiological function.
Common predators of this species include the common leopard, Panthera pardus, and the snow leopard, Panthera uncia. Other potential threats include wolves, Canis lupus; dholes, Cuon alpinus, a wild dog that hunts in packs; and golden jackals, Canis aureus, that may prey on young or sick langurs. These, however, are easily avoided by healthy adults who can seek refuge in the safety of their trees.
Generally, when langurs encounter predators, they emit alarm calls, display aggression, or flee. In areas where Nepal gray langurs live near rural human settlements, interactions with people and domestic dogs have increased. Langurs are harassed or attacked, either as bushmeat targets or to deter crop raiding. This has created a “landscape of fear,” adding another layer of pressure for langurs to navigate.
In response, adult langurs in these areas have begun to counterattack domestic dogs, a relatively recent behavioral shift. In contrast, langurs living in forests away from villages rarely show aggression toward dogs, likely because they have not experienced the same persistent threat. These counterattacks are most often initiated by high-ranking males with a nearby family troop. In contrast, lower-ranking males rely more on alarm calling. Lone males typically flee or freeze rather than engage. This emerging pattern highlights the behavioral flexibility of Nepal gray langurs. It underscores their capacity for problem-solving and social decision-making in changing environments.
Their tails point forward, unlike their southern cousins. Some scientists believe this might be a cultural behavior, that is, a learned behavior passed down through generations.
Black Himalayan bears take advantage of Nepal gray langurs’ eating habits by snacking on acorns that langurs accidentally drop while foraging.
The Abominable Snowman (or ‘Yeti’) might be none other than the elusive Nepal gray langur!
The Nepal gray langur lives in cohesive multi-male, multi-female groups where social structure is largely governed by physical condition and age. They are social animals that live in groups of about 4-47 individuals, with 1-4 adult males. All-female groups and solitary males have also been spotted. Their home range is quite extensive, around 3,150–31,380 acres (1,275–12,700 ha).
Nepal gray langurs begin foraging early in the morning, with peak feeding activity between 9 a.m. and noon. They rest during the hottest parts of the day. During this period, juveniles play, while adults engage in social behaviors such as allogrooming (cleaning each other’s fur) and autogrooming (self-cleaning). Adults may also engage in geophagia as their food is being digested.
Across all categories — adult males, adult females, subadults, and juveniles — resting accounts for up to 50% of the day, with juveniles resting slightly less than adults. Around 30% of their day is devoted to moving, followed by feeding, which takes 40% of their day. Females tend to rest more compared to males, while youngsters rest significantly less. Social behavior, including grooming, takes up to 20% of juveniles’ day, while adults spend around 10% of their day interacting with others. Females groom other members of the group more often than males. Playful behavior, instead, is only typical of juveniles or subadults, with adults hardly ever playing.
Male dominance relationships are central to troop dynamics, particularly in relation to access to females and reproductive opportunities. Their social dominance follows a chain-of-command logic with an alpha ruling over different betas, who themselves follow their own internal hierarchy.
Typically, young adult males (7–9 years of age) hold the highest ranks. Subadult males rank the lowest, while older males often decline in rank as physical strength wanes. In addition to age, physical condition plays a crucial role: males in better shape tend to have higher ranks. Coalitions among males are absent — every langur for himself.
There are two main tactics males use to become alpha:
Immigration — This is the most common route. A male from outside the group enters and, through aggressive encounters, overthrows the current alpha. This high-risk tactic is favored by younger, stronger individuals. They tend to have long-lasting reigns.
Residency or Succession — Less frequently, a long-time resident male ascends to alpha status when more dominant males emigrate or die. There’s less physical risk, but it is also less effective. This leads to shorter reigns and fewer offspring.
Some males are pretty tough rulers. These “hyper-aggressive” individuals actively stalk, chase, and attack other group members, creating an atmosphere of high tension. While they tend to maintain their alpha status longer, they are not notably more successful in siring offspring. Aggression may secure dominance, but not necessarily reproductive success.
Females tend to coexist peacefully and form their own hierarchy. Groups are mostly composed of mothers and their daughters, which creates naturally strong, kin-based bonds. Older females are typically higher ranking and have produced much of the troop’s offspring, giving them the widest network of social relationships.
Subadult females groom others more frequently and act as the “glue” of the social network. Grooming reinforces bonds, and the groomer can gain social favor from the individual being groomed.
The Nepal gray langur is an exceptionally vocal primate. Vocalizations play a fundamental role in the daily life of this species, particularly due to the challenges of living in large social groups that often travel across steep Himalayan forests.
Adult males are the most frequent callers, their calls usually being louder and more forceful. Unlike the southern Indian or plains-dwelling langurs, the Nepal gray langur does not emit the iconic “whoop” call; instead, they produce a sound that resembles a “Wao” or “Ua”.
Another call commonly heard in high-tension situations is the harsh bark, produced when rival males approach or when a predator is spotted nearby. These harsh, sudden vocalizations often come alongside visual displays, such as aggressive postures or teeth grinding. During tense male interactions, males will also emit pant barks (short, breathy expulsions of air) and rumble screams (lower-pitched, resonating calls that may escalate in intensity). Teeth grinding is often seen in tandem with these calls and functions as a visual warning.
Females, while generally quieter than males, are far from silent. They produce a variety of more melodic and higher-pitched sounds. In social contexts involving tension, playful interactions, or contact with infants, females may emit warbles, squeals, shrill calls, and whistles. These vocalizations are used to signal discomfort, establish boundaries, or draw attention, particularly when caring for young. During close social bonding, females may produce contact tremolos, which are low, rhythmic sounds used to reinforce social cohesion. During grooming, they chatter their teeth. When separated from the group, females also emit isolation peeps to reestablish contact, especially with their offspring.
Subadult langurs produce a similar vocal range to females. Warbles and squeals are common among them, especially during games or when reacting to sudden rebuffs. These vocalizations serve as both social signals and emotional outlets, reflecting their still-developing status within the group.
Juveniles are among the most vocal individuals in the troop. Their calls are frequent and highly variable. In addition to whistles and squeals, juveniles emit shrieks and warbles, especially when playing, experiencing distress, or being separated from caregivers. Their calls are often rapid and repetitive, drawing the attention of adults nearby.
Adult males, adult females, and juveniles all emit alarm calls when spotting nearby threats.
Even infants possess a unique vocal repertoire. One of the most distinct infant calls is the wail, a drawn-out, plaintive sound associated with weaning or moments of separation from the mother. During group reunions or when startled, infants may emit hiccup-like calls. Perhaps most notable is the milk grumble, a soft, rhythmic sound produced during nursing sessions. This vocalization appears to strengthen the bond between mother and infant and is not heard in any other context.
Grooming plays an important role within Nepal gray langur groups. It functions as a social glue, used to maintain and strengthen social bonds. It’s a highly effective means of communication.
A single top-ranking male, the so-called alpha, tends to monopolize most of the mating opportunities, while subordinate males have significantly reduced reproductive success and have to be sneaky about it. Nearly 74% of the infants are fathered by the alpha male, one of the highest levels recorded among primates. Mothers give birth to a single infant, rarely twins, and their gestation period is around six months. They usually give birth between July and September. Infants are born helpless and need the full support of their mothers to become strong and independent. They’re usually weaned when they turn two, which is when females might become pregnant again.
Nepal gray langurs breed seasonally. Timing an alpha’s rise to power strongly affects his reproductive success, as males who become alpha during the non-breeding season may end up holding a high rank without fathering any offspring.

As an herbivore and leaf-eating mammal, the Nepal gray langur contributes to life in the forest in several ways. Firstly, by consuming leaves and pine needles, Nepal gray langurs prune trees and allow new, healthier vegetation to grow. Their pruning activity also helps sunlight reach the forest floor, contributing to new growth. They also feed on seeds and fruits, thus contributing to forestation while foraging across their home range.
More interestingly, the Nepal gray langur has been observed foraging together with other species who benefit from their “messy” feeding habits. For example, researchers have observed Himalayan black bears, Ursus thibetanus, stationed under the trees where Nepal gray langurs feed to catch any acorns that are accidentally dropped. The Himalayan black bear, an omnivore, is a potential predator of this species, which makes this association even more interesting.


The Nepal gray langur is classified as Least Concern by the International Union for Conservation of Nature (IUCN, 2015), appearing on the IUCN Red List of Threatened Species. This species is widespread and is not currently facing serious threats that may lead to rapid population decline. However, their home range is shrinking due to human activity, which might seriously affect their numbers in the near future. Current threats include logging, fires, expansion of human activities and habitations, developmental activities, and war. Hunting poses a significant threat as well: in Tibet, this species is used in traditional native medicine, while in Nepal, their cured meat is considered a delicacy.
Nepal gray langurs are listed in Appendix I of the Convention on International Trade in Endangered Species (CITES), an international agreement between governments whose goal is to ensure that international trade in specimens of wild animals and plants does not threaten their survival. This species inhabits several protected areas within the Himalayan range, with populations in at least ten conservation zones across South Asia.
In India, the species can be found in Chail Wildlife Sanctuary, Changthang Wildlife Sanctuary, Dachigam National Park, Hemis National Park, Khangchendzonga National Park, and Karakoram Wildlife Sanctuary. In Nepal, they seek refuge in Langtang National Park, Makalu Barun National Park, and Everest National Park. Finally, in Pakistan, they live in the Manshi Wildlife Sanctuary, one of the few known localities in the country to support Himalayan langurs.
- Abominable Snowman! (2010) The Santa Barbara Independent. Available at: https://www.independent.com/2010/12/28/abominable-snowman/
- Arekar, K., Sathyakumar, S. and Karanth, K.P. (2021) Integrative taxonomy confirms the species status of the Himalayan langurs, Semnopithecus schistaceus Hodgson, 1840. Journal of Zoological Systematics and Evolutionary Research, 59(2), pp.543–556.
- Bhaker, N.R., Rajpurohit, D.S. and Rajpurohit, L.S. (2004) Vocalization in Hanuman langur, Semnopithecus entellus, around Jodhpur, Rajasthan. Uttar Pradesh Journal of Zoology, pp.227–233.
- Borries, C., Perlman, R.F. and Koenig, A. (2017) Characteristics of alpha males in Nepal gray langurs. American Journal of Primatology, 79(7), e22437.
- Franklin, S. (n.d.) Seasonal variation in the diet of Himalayan grey langur (Semnopithecus ajax) in Machiara National Park, Azad Jammu and Kashmir, Pakistan.
- Gray langur (n.d.) Wisconsin National Primate Research Center. Available at: https://primate.wisc.edu/primate-info-net/pin-factsheets/pin-factsheet-gray-langur/
- Himalayan langur (n.d.) Nepal Desk. Available at: https://nepaldesk.com/mammal/himalayan-langur
- Minhas, R.A., Ahmed, K.B., Awan, M.S. and Dar, N.I. (2010) Habitat utilization and feeding biology of Himalayan grey langur (Semnopithecus entellus ajax) in Machiara National Park Azad Kashmir (Pakistan). Zoological Research, 31(2), pp.177–188.
- Monaco, E.T., Borries, C., Nikolei, J., Chalise, M.K., Ganzhorn, J.U., Wesche, K. and Koenig, A. (2019) The function of geophagy in Nepal gray langurs: sodium acquisition rather than detoxification or prevention of acidosis. American Journal of Physical Anthropology, 168(1), pp.170–179.
- Nautiyal, H. (2015) Life in the extreme: time-activity budgets and foraging ecology of central Himalayan langur (Semnopithecus schistaceus) in the Kedarnath Wildlife Sanctuary, Uttarakhand, India.
- Nautiyal, H. (2020) Behavioral ecology of the central Himalayan langur (Semnopithecus schistaceus) in a human-dominated landscape: multi-species interactions and conservation implications. Kyoto University.
- Nautiyal, H., Huffman, M.A. (2018) Interspecific feeding association between central Himalayan langurs (Semnopithecus schistaceus) and Himalayan black bears (Ursus thibetanus) in a temperate forest of the Western Indian Himalayas. Mammal Study, 43(1), pp.55–60.
- Nautiyal, H., Tanaka, H. and Huffman, M.A. (2023) Anti-predator strategies of adult male central Himalayan langurs (Semnopithecus schistaceus) in response to domestic dogs in a human-dominated landscape. Primates, 64(3), pp.361–379.
- Nautiyal, H., Mathur, V., Gajare, K.H., Teichroeb, J., Sarkar, D. and Diogo, R. (2024) Predatory dogs as drivers of social behavior changes in the central Himalayan langur (Semnopithecus schistaceus) in agro-forest landscapes. Biology, 13(6), p.410.
- Nautiyal, H., Romano, V., Tanaka, H. and Huffman, M.A. (2024) Female social dynamics as viewed from grooming networks in the central Himalayan langur (Semnopithecus schistaceus). American Journal of Primatology, 86(8), p.e23655.
- Perlman, R.F., Borries, C. and Koenig, A. (2016) Dominance relationships in male Nepal gray langurs (Semnopithecus schistaceus). American Journal of Physical Anthropology, 160(2), pp.208–219.
- Sharma, N., Huffman, M.A., Gupta, S., Nautiyal, H., Mendonça, R., Morino, L. and Sinha, A. (2016) Watering holes: the use of arboreal sources of drinking water by Old World monkeys and apes. Behavioural Processes, 129, pp.18–26.
- Singh, M., Kumara, H.N., Yongcheng, L., Chetry, D. and Kumar, A. (2020) Semnopithecus schistaceus. The IUCN Red List of Threatened Species 2020, e.T39840A17942792.
Original text by Mirko Cerrone (May 2020); revised by Acima Cherian (April 2026)
