Malbrouck, Chlorocebus cynosuros
MALBROUCK
Chlorocebus cynosuros

Geographic Distribution and Habitat
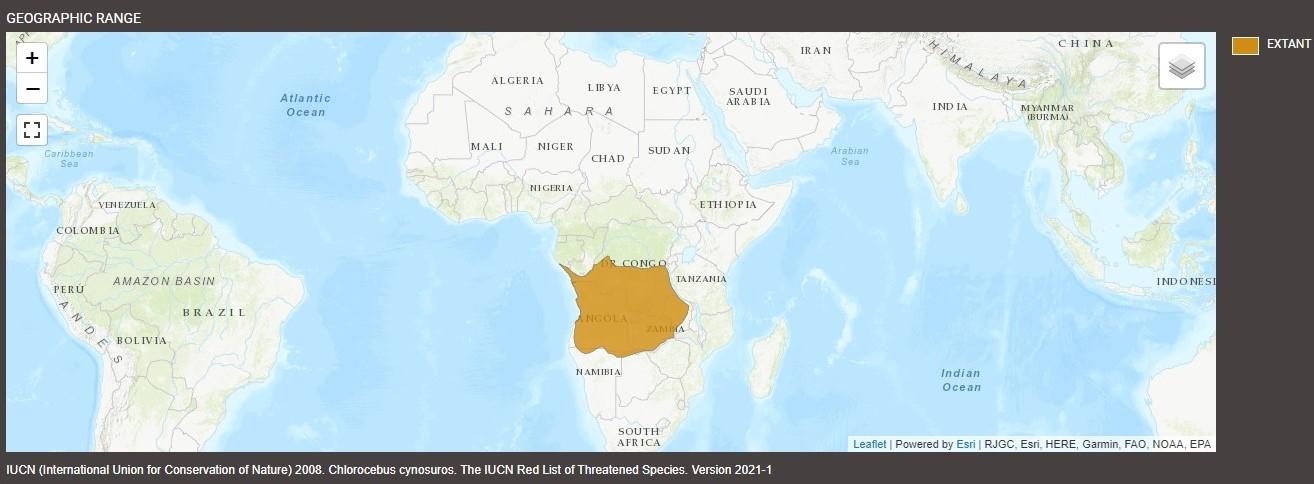
Native to central and southern Africa, malbrouck monkeys (Chlorocebus cynosuros) are medium-sized primates that inhabit a wide range of wooded and open environments. Malbroucks carry many common names, including African green monkey and savanna monkey, though they are often misidentified as “vervets” or “grivets”—terms that technically refer to related species (Chlorocebus pygerythrus and Chlorocebus aethiops, respectively).
Indigenous to Angola, western Zambia, southern Democratic Republic of Congo, northern Namibia, and northern Botswana, their populations possibly extend into southern Gabon. The Zambezi River basin marks their southernmost boundary, while the Luangwa River serves as a consequential biogeographic barrier, maintaining the evolutionary separation between western populations from eastern species like vervets, which do not occur west of the East African Rift or the river.
The species is distinguished by its ecological plasticity. Malbroucks can adapt to most habitats, although they thrive in what ecologists call an “optimal habitat zone,” and must be close to a water source. Textbook generalists, these monkeys need conditions that are “just right,” without being extreme in any direction. This explains their presence in specific regions of Africa while remaining absent from others that might seem similar.
Preferring miombo and mopane woodlands, they inhabit areas where deciduous trees form a loose, open canopy with herbaceous layers of grasses and forbs. Yet these monkeys are just as comfortable in marshy forests, riverine corridors, montane scrub, and even human-modified areas. As pristine habitats continue to shrink across their range, many malbroucks have successfully adapted to secondary forest, agricultural edges, and village peripheries, which often puts them in direct conflict with humans.
Most African monkeys are limited to low- or mid-elevation habitats, where temperatures are stable and vegetation stays plentiful year-round. Malbroucks, however, can endure colder environments exceeding 13,100 feet (4,000 m), though most groups are found below 5,200 feet (1,600 m). Residential temperatures in their habitats hover between 18–24°C (64–75°F) year-round, rarely dropping below 5°C (41°F).
Malbrouck monkeys have spent the last two centuries caught between competing taxonomic philosophies, their classification repeatedly modified as scientists debate how best to organize African primate diversity. Taxonomists don’t always agree on what defines a species. Some downplay subtle differences between populations, chalking them up to local variation and grouping similar forms into a single species with regional subspecies in a practice called “lumping.” Others emphasize distinctions in appearance, behavior, or genetics as grounds for full species status (“splitting”).
Malbrouck monkeys (Chlorocebus cynosuros) belong to the Cercopithecidae family of catarrhine primates, which are “dry-nosed” primates that include African-Eurasian (formerly Old World) monkeys, apes, and humans.
Cercopithecidae contains two subfamilies: Cercopithecinae (cheek-pouch monkeys) and Colobinae (leaf monkeys). Malbroucks fall within Cercopithecinae, specifically the tribe Cercopithecini, which contains six distinct genera:
- Allenopithecus (Allen’s swamp monkey), 1 species
- Miopithecus (talapoins), 2 species
- Erythrocebus (patas monkeys), 3 species
- Chlorocebus (African green monkeys), 6 species
- Allochrocebus (terrestrial guenons), 3 species
- Cercopithecus (arboreal guenons), 25 species
Within Chlorocebus, malbroucks share their genus with five other species distributed across sub-Saharan Africa: grivet (C. aethiops), Bale monkey (C. djamdjamensis), vervet (C. pygerythrus), green monkey (C. sabaeus), and tantalus monkey (C. tantalus).
First described in 1786 as Simia cynosuros, malbroucks were initially classified in the catch-all Simia genus that housed everything from baboons to lemurs. Early Linnaean taxonomy relied on superficial traits to differentiate species, often grouping them simply because they didn’t obviously fit elsewhere. In the mid-19th century, some experts reclassified malbroucks into Cercopithecus, joining other tailed primates in a more organized framework, and Simia was officially suppressed in 1929.
African green monkeys display nearly identical morphology, have overlapping behaviors, and frequently hybridize where their ranges meet. Therefore, in absence of extensive behavioral data, genetic tools, and long-term field studies, especially from remote parts of sub-Saharan Africa, many in the Chlorocebus genus were regarded as regional variants of a single polytypic species.
From the 1950s through the 1990s, all vervet-like monkeys were consolidated under the umbrella species Cercopithecus aethiops because, in addition to incomplete sampling of the species, phenotypic (“physical traits”) similarities were treated as a proxy for shared ancestry. Malbroucks were consequently relegated to subspecies C. a. cynosuros under the dominant lumping philosophy that guided the field at the time.
Once this lumped framework was established, it created a self-reinforcing system where new findings were interpreted through outdated terminology. To this day, many reputable older sources incorrectly identify malbroucks as vervets or grivets.
Skull morphometrics and DNA studies in the 1980s through the 2000s revealed that vervet-like monkeys formed a distinct evolutionary lineage separate from true Cercopithecus species. In 2001, the genus Chlorocebus was resurrected, elevating the malbrouck to full species status. Subsequent molecular studies in the 2010s validated this revision, and modern taxonomy treats Chlorocebus cynosuros as a monotypic species.
Size, Weight, and Lifespan
Measuring 16-24 inches (40-60 cm) from head to rump, malbroucks are equipped with whip-like tails that add another 20-28 inches (50-70 cm), often exceeding the length of head and body combined. Within the guenon family, they are larger than vervets, but smaller than imposing baboons (genus Papio).
Sexual dimorphism, or the physical difference between the sexes other than those that define their gender, is less extreme than in some other primates. Males can be 30-50% larger than females, weighing 9-17 pounds (4-8 kg) compared to females’ 7-11 pounds (3.5-5 kg). Broader chests, larger canines, and stronger brow ridges further distinguish males.
Because political instability across much of their range makes long-term field studies dangerous, and because malbroucks were treated as subspecies for decades, their biology and behavior are woefully understudied. Researchers, therefore, infer from better-studied Chlorocebus species that malbroucks likely live 12–18 years in the wild and up to 25–30 years in captivity.
Appearance
Unlike the thick, interlaced canopy of tropical rainforests, the wide, flattened pagoda crowns of the miombo woodlands seldom touch, leaving ample gaps between trunks. Because their environment doesn’t offer the luxury of close-set branches, malbroucks are built lean and athletic to be as agile on the ground as they are in trees.
Their narrow torsos flow into long, proportional limbs designed for leaping and sprinting across sun-baked clearings. Behind them, their elongated, non-prehensile tails arch and counterbalance, helping them move with control when they must move swiftly.
Most newborns begin life with soft, dark brown fur, which matures into a grizzled gray-green and warm beige coat that blends into the dry foliage and substrates of their habitat. Creamy white undersides contrast with olive drab flanks, while their limbs darken into dusky hands and feet that help obscure the monkey’s outline in shadows cast by scattered trees.
Pale, blotchy faces freckled with upward-sweeping cheek whiskers distinguish them from other Chlorocebus species, whose whiskers typically point downward from jet-black faces. They have short, dry noses with closely-set nostrils, reflecting their reduced reliance on smell compared to their more ancestral primate relatives. Unlike the moist rhinarium (long, wet-nose) of lemurs and galagoes, their flatter noses exemplify the evolutionary shift toward vision that occurred when the haplorrhine lineage split from strepsirrhines approximately 65 million years ago.
Forward-facing brown eyes sit beneath prominent cream-colored brows that give them a quizzical furrow. Though they have lost ancestral traits like the tapetum lucidum (the reflective eye layer that aids night vision), day-active malbroucks have evolved trichromatic color vision. They can see the full spectrum of colors that humans see, including reds, greens, and blues, to more effectively find coveted food like fruit in the patchy, competitive savanna woodlands.
Facial expressions and postures help maintain group cohesion and coordinate movements, but these nonverbal cues are easily lost in the visual noise of moving grass or shadowed tree bark. White tufts of fur around the brows, cheeks, and jaw create a high-contrast frame that makes individual expressions more visible to others in the patch-and-pall sunlight.
Malbroucks are often seen with inflated cheeks. African-Eurasian monkeys possess expandable buccal sacs that function as portable pantries during foraging expeditions. Foraging is dangerous as it leaves an individual exposed as they concentrate on selecting and consuming food. Cheek-pouches let them quickly gather provisions before retreating to safer locations for unhurried processing.
Both sexes bear the bluish abdominal skin that marks them as members of the Chlorocebus lineage, but males have lapis blue scrotal skin and red penises that vividly broadcast their sexual maturity, health, and rank. Researchers have found that those with higher androgen levels tend to have more blue scrotal coloration, while higher-ranking males display more luminant and saturated red penises.

Diet
Among Africa’s most mercurial biomes, the malbrouck’s habitat cycles through drought and deluge, where long, parching dry spells (May-October) are punctuated by the wet season’s (November-March) brief, turbulent rains. Even then, the timing and amount of rainfall are fiercely variable from year to year, and within the same region. A single delayed rain can postpone flowering, fruiting, and leafing by weeks or months, making it difficult for animals to rely on consistent resources. Such an ecosystem can be unfriendly to animals with specialized diets; the species that survive here tend to be flexible, opportunistic, and metabolically prepared for change.
Anatomically, these monkeys are equipped to process drastically different types of food across seasons, without incurring trade-offs that many other species suffer. Simple, single-chambered stomachs are partnered with complex colons and caeca packed with beneficial microbes that can digest denser plant materials when preferred foods disappear. Their cheek pouches have the same volume as their stomachs and produce saliva rich in amylase enzymes that break down starches into digestible sugars while the monkeys move between feeding sites. This is especially useful for foods like acacia seeds, which need extra time to soften before reaching the stomach.
Broad, low-cusped molars grind through thick leaves and pods, while pointed premolars slice fruit skin and crack shells. The elongated canines in males, though more so wielded during tense negotiations, can extract stubborn invertebrates from bark crevices. Even their enamel thickness strikes an optimal balance: durable enough for tough materials yet not so heavy as to waste energy when consuming softer foods.
Much of what we know about the malbrouck’s diet comes from detailed studies conducted when they were still considered a subspecies. Formally, they are classified as seasonal frugivores and folivores that favor soft, sugar-rich fruits and flowers comprising 60–80% of their diet during wet season abundance. They prize wild fruits like guava (Psidium guajava), mango (Mangifera indica), and avocado (Persea americana), alongside cultivated crops they encounter near human settlements. When succulent fruits vanish in the dry season, their omnivory expands to include seeds, gum, insects, eggs, chicks, and lizards. They also raid agricultural crops, such as beans, peas, and maize, and aren’t above scavenging human refuse or pilfering kitchen scraps when opportunities arise.
Malbroucks have learned to anticipate peak ripeness and travel great distances to access specific fruiting trees. They arrive just as the fruits’ sugar content is at its highest, but before competing frugivores can monopolize the resource. This knowledge is passed down through social learning, as older individuals learn patterns through experience, and younger monkeys learn by observing.
Within the same troop, the types of food individuals eat can also be affected by social rank. Dominants seize the most productive feeding sites, forcing subordinates to either wait their turn or seek alternative food sources. Cheek pouches are tactically handy amid such competition; a quick-acting subordinate can stuff a substantial amount of food into their cheeks before ceding a site to an approaching dominant.
Behavior and Lifestyle
Malbroucks exhibit the behavioral flexibility that characterizes successful Cercopithecinae. Much of what we understand about their daily lives comes from studies of vervets and grivets.
They are diurnal, semi-terrestrial, and semi-arboreal primates who split their time between the ground and the trees, using the canopy to evade predators, sleep, and reach otherwise inaccessible food. Terrestrial quadrupeds by default, malbroucks walk with their palms and soles flat to the ground in a plantigrade stance (with their entire foot touching the ground or other surface). Using a diagonal sequence gait, they lift a hand and the opposite foot forward in steady alternation. They can rear up on their hindlimbs to scan for threats, though it’s a temporary, facultative behavior (one they can choose or not, depending on circumstances)—their skeletal structure isn’t suited for prolonged upright postures.
While they can move in trees, their leaping is modest and typically occurs as brief bounds between supports, rather than the powerful, ballistic jumps of arboreal specialists, like colobus monkeys. They climb using all four limbs, aided by clawless, pentadactyl hands (with five digits) lined with dermal ridges for grasping branches.
Studies of related species suggest they follow typical primate activity patterns: morning foraging, midday rest, and afternoon feeding sessions. However, the seasonality of food influences their daily ranging behavior. During wet season abundance, troops keep smaller home ranges and are pickier about what they eat. As dry season scarcity intensifies, they increase daily travel distances to search for lower-quality foods.
Social rank determines access to preferred feeding sites and water sources. Dominant females and their offspring typically feed first at productive trees, while subordinates either wait for access or station themselves at alternative food spots. Established matrilines (groups of related females) also drink first, while others maintain careful distance until the hierarchy permits their approach.
At dusk, troops retreat to communal roosting sites, showing documented preference for fever trees (Acacia xanthophloea) when available. These thorny trees provide protection from terrestrial predators while their structure accommodates multiple individuals. Sites are typically positioned near the day’s final feeding areas and within reasonable distance of anticipated morning foraging locations, reducing their vulnerability to predators.
Chlorocebus monkeys beautifully demonstrate semantic communication in primates, where different signals represent distinct objects in the environment. Malbroucks are believed to produce distinct alarm calls for specific predators that elicit behavioral responses within the troop, as observed in vervets.
When aerial predators, like martial eagles (Polemaetus bellicosus) or Verreaux’s eagle owls (Ketupa lactea), are spotted, the observer emits low-pitched, staccato grunts. Hearing these, nearby members look skyward and either flee into nearby brush or crouch low in cover. They emit high-pitched “chutters” in response to snakes such as the African rock python (Python sebae), prompting individuals to scan the ground. Leopards (Panthera pardus) and dogs (Canis familiaris) are met with a series of short, tonal calls, in which case, grounded monkeys quickly ascend trees.
Malbroucks are among the world’s highest-dwelling primates.
Baby malbroucks in Angola are born with silver coats vs. the typical browns.
The male’s blue scrotal skin results from the Tyndall effect, where light scatters through unusually organized collagen fibers in the skin.
They live in polygamous troops ranging from 8 to over 50 individuals, though most groups contain around 25 members. Adult males and females exist in roughly equal numbers alongside their offspring. Malbroucks practice female philopatry, where daughters stay with their birth groups throughout their lives. This permanence allows females to forge enduring bonds with mothers, sisters, and aunts that stabilize hierarchies across generations.
The malbrouck monkey’s social structure is assumed from the typicality of vervets, African green monkeys, and grivets. Rank likely passes from mother to daughter, and high-ranking females will inherit the best resources and alliances. For primates, grooming is excellent social currency for those born into subordinate positions. Lower-ranking females may strategically initiate grooming sessions with dominants, seeking tolerance at feeding sites and protective coalitions for themselves or their offspring.
Characteristics of polygamy, as well as inferences made from other species, suggest that male rank, in contrast, is hard-earned and impermanent. To avoid inbreeding and access better mating opportunities, adolescent males disperse from their natal groups in search of troops to join. The precise details of male immigration and how hierarchies are established in malbroucks haven’t been documented.
Takeovers in other savanna monkeys predictably incite group-wide tension that culminates in aggression and physical fights. The same is presumed for this species. Dominant females chase immigrants, recognizing the threat they pose to the group’s stability and their own vulnerable offspring. New alpha males may kill infants fathered by their predecessors to force females back into estrus, as seen in related species.
Males technically outrank females and can interrupt disputes, commandeer feeding sites, or intimidate rivals away from preferred resting spots. The alpha male enjoys preferential access to females. He patrols the group’s periphery, scanning for threats or competing males. Yet despite this formal dominance, practical authority belongs to senior females, the troop’s true social anchors, who determine where groups feed, when they rest, and how long they remain in particular areas.
They may have home ranges of approximately 0.19 to 0.77 square miles (0.5 to 2 km²), defending core territories containing essential resources against neighboring troops, as comparable Chlorocebus species do. If they follow patterns seen in vervets and grivets, they would establish zones of tolerance, where overlapping ranges allow different groups to forage at separate times or practice mutual avoidance. Confrontations in similar monkeys don’t often escalate to chasing or physical contact, though territorial disputes occasionally erupt into brief scuffles involving grabbing, slapping, or biting.
Malbroucks coexist alongside a plethora of mammals, birds, reptiles, and even domestic animals. Ungulates like impala (Aepyceros melampus), bushbuck (Tragelaphus scriptus), and common duikers (Sylvicapra grimmia) graze or browse in the same areas, and the monkeys sometimes benefit from their presence. For instance, grazers may stir up delectable insects when they move across the grassland, or detect a predator unseen by the malbrouck troop.
Baboons, on the other hand, particularly olive baboons (Papio anubis) and yellow baboons (Papio cynocephalus), are formidable competitors known to forcibly expel malbroucks from preferred feeding sites in regions where both species co-occur. Some baboons are even documented as opportunistically predating on smaller monkeys.
As haplorhines (dry-nosed primates), malbroucks probably communicate primarily through facial expressions, body language, and learned vocal signals rather than relying on scent like strepsirrhines (moist-nosed prosimian primates). While direct research on malbrouck communication remains limited, observations of related species suggest how these monkeys likely interact with one another.
Stiff-legged walking, chest-puffing, and raised tails are declarative assertions of dominance, deployed when patrolling feeding sites or confronting newcomers. When tensions escalate, subordinates resort to “fear grimaces,” flattening their ears, averting their gaze, and baring their teeth in desperate attempts to defuse mounting aggression. Lip-smacking and gentle, audible clicks impart friendliness and solicit contact, like grooming.
In savannas, there are fewer physical barriers to block sound, but the distances between individuals are much greater when they spread out to forage. Malbroucks have likely evolved vocalizations that are louder, longer, and lower in frequency than those of forest relatives. It’s not enough for a call to simply reach another monkey; the receiving animal needs to distinguish between different types of calls (alarm, location, social) and identify who’s calling. The acoustic properties have to preserve enough detail for these distinctions to remain clear across distance.
Grunts are low-amplitude vocalizations that are used in a variety of contexts. A malbrouck may find himself socially rebuffed with a short grunt of annoyance, while louder contact calls announce their passive presence to another group.
Infants are born with the vocal apparatus to imitate the screams, whrrs, and grunts of adults, but what isn’t innate is an understanding of what those calls mean or when they’re appropriate. That knowledge is acquired through social experience, and the learning curve reveals itself in juveniles’ mistakes. Adults don’t teach youngsters directly; instead, they provide social feedback through differential reinforcement. A juvenile’s incorrect or poorly timed call may be ignored or met with a delayed reaction, allowing the young monkey to gradually discern which vocalizations prompt a full group response.
Direct data on malbrouck reproductive behavior doesn’t exist, but it can be presumed from other species in their genus, like vervets and green monkeys.
When Central Africa’s rains arrive, the savanna-woodlands burst with energy-dense fruits. Like many primates living in seasonal environments, malbroucks probably time reproduction with this abundance. Fruits fuel not just the caloric demands of females’ pregnancy and nursing, but also males’ sperm production and the exhausting business of sexual competition.
Females reach sexual maturity around 3 years of age, while males begin producing sperm at similar ages but don’t hit their competitive stride until 5-6 years. Like other Chlorocebus monkeys, they are polygamous: dominant males choose the most fertile females, while subordinate males resort to sneaking around or forcing unwanted encounters. In these multi-male, multi-female groups where females mate with several partners, the strongest males may win the obvious battles, but female promiscuity ensures every male she mated with has a chance at parenthood.
Pregnancy stretches about 5.5 months, timing births for the early dry season when food is still plentiful but competition for it eases. Females typically produce one infant yearly, though twins are a rare surprise. Subsequent births average 12-24 months, depending on environmental stress, nursing duration, and how quickly mothers resume ovulating after weaning.
Malbrouck babies arrive fully furred, eyes open, and ready to cling to their mother’s belly, later graduating to riding on her back. New mothers become security blankets, maintaining constant contact during those vulnerable first weeks. Most wean between four and six months, though some stressed juveniles will seek nursing sessions until they gain social confidence around their first year.
Males don’t usually participate in raising the young, which is standard operating procedure in polygynous species where paternity is uncertain. This uncertainty actually protects baby malbroucks: males avoid killing infants because they might murder their own offspring. Occasionally, a male tolerates youngsters climbing on him, sitting nearby, or even playing without reacting aggressively or chasing them away. It’s not parenting, exactly, but this social tolerance helps young monkeys decode group politics and practice social skills safely. Siblings and nulliparous females (adult females who haven’t yet had offspring) are the most active alloparents, assisting with grooming, carrying, or protecting the infant to improve their chances of survival, as well as rehearsing their own future parenting roles.

Miombo woodlands are populated by Msasa (Brachystegia spiciformis), Bloodwood (Pterocarpus angolensis), wild loquat (Uapaca kirkiana), and other leguminous trees, whose open-canopy structure and symbiotic relationships with nitrogen-fixing bacteria support over 8,500 species of plants. Many of these trees are endozoochorous; they attract animals to consume their fruits and deposit seeds away from the parent plant through their feces.
Malbroucks are dietary generalists who are very mobile across their range, especially when they are after a specific food. They consume a variety of fruit, even those whose seeds are too large or tough for smaller animals to handle. If a seed falls under the parent tree, it is less likely to germinate as it competes with the parent for light, water, and nutrients, and is besieged by insects and rodents that concentrate around the tree.
Since malbroucks are adaptable and tolerate human disturbance better than many other frugivorous mammals, they may be one of the few remaining seed dispersers for certain tree species in the increasingly fragmented habitats.


The malbrouck monkey is classified as Least Concern by the International Union for Conservation of Nature (IUCN, 2019), appearing on the IUCN Red List of Threatened Species. When last assessed in 2019, populations were considered stable throughout their range. However, for lesser-studied species like the malbrouck, “stable” may simply reflect inadequate monitoring rather than true ecological security. Currently, no comprehensive estimates exist for their total population in the wild, nor have reliable, large-scale studies been conducted on this species.
The “Least Concern” designation does not render these monkeys invulnerable. Even common species remain susceptible to local extirpation due to hunting, habitat loss, and human-wildlife conflict. Although not a major target of the bushmeat trade, malbroucks are occasionally hunted for subsistence in parts of their range.
The primary threat facing malbroucks epitomizes the habitat destruction and fragmentation that many primates throughout the continent endure. Central Africa experiences deforestation at a rate nearly double the global average. This relentless conversion of forest into agricultural land, along with logging and development, continues to shrink the landscapes that malbroucks and other less adaptable animals depend on.
The dietary flexibility and increased terrestriality seen in the Chlorocebus genus have allowed many of its members to withstand rapid environmental changes. Still, such disruptions reshape how they forage, socialize, and move through their home ranges, introducing pressures that frequently force wildlife into direct confrontation with human communities. Paradoxically, their adaptability and tendency to thrive near humans also increase their exposure to conflict-related mortality.
Malbroucks are notorious crop raiders, particularly in Angola and Zambia, where they target maize, sorghum, and various fruits. This behavior has earned them a reputation as agricultural pests, leading to retaliatory killings through poisoning, shooting, and trapping. Opportunistic and intelligent, they also plunder garbage and enter homes, endearing them less to locals. In some areas, organized “drives” to round up and kill these monkeys have been documented, though these practices are fortunately less common than they once were. Juveniles and infants are sometimes captured for the illegal pet trade, while others are killed by domestic dogs or hit by vehicles while crossing roads.
Beyond physical danger, the close evolutionary relationship between humans and primates makes both susceptible to many of the same pathogens, facilitating zoonotic disease transmission. When monkeys forage in human settlements and agricultural areas, they can carry tick-borne pathogens such as Anaplasma phagocytophilum, Rickettsia species, and Babesia, bringing these microbes into closer contact with people—and potentially picking up new pathogens from domestic animals or humans. Diseases like tuberculosis and measles can also spread from humans to primates, sometimes triggering severe outbreaks in wild populations.
Malbrouck monkeys are listed in Appendix II of the Convention on International Trade in Endangered Species (CITES), an international agreement between governments whose goal is to ensure that international trade in specimens of wild animals and plants does not threaten their survival. This designation recognizes that while not immediately threatened, the species could become so if trade is not regulated.
In practice, malbroucks receive little direct conservation attention. Their wide distribution, ecological flexibility, and close resemblance to other Chlorocebus species make them less visible to researchers, conservationists, and funders compared to more charismatic or imperiled primates in the region. Since they are regarded as pests, many locals state they’d rather the government shrink their numbers than conserve them.
They occur in several protected areas, indirectly safeguarded in strongholds such as Luando National Park (Angola), Chobe National Park (Botswana), Kafue National Park (Zambia), and Etosha National Park (Namibia). The quality of that protection greatly depends on the country, however. Enforcement in Angola, for instance, has been weakened by insufficient resources, land scarcity, and civil conflict that left many parks unmanaged.
Beyond national parks, rehabilitation centers across southern Africa, some affiliated with the Pan African Sanctuary Alliance (PASA), care for confiscated monkeys, including Chlorocebus. While these centers focus on re-release, they also act as intelligence hubs for mapping trafficking routes and identifying repeat offenders.
Still, the limitations of conservation that rely on boundaries and punitive measures have been made clear by continued decline of Africa’s wildlife. Fines, prosecutions, and patrols may deter opportunistic poaching, but they do little to address why people turn to wildlife in the first place.
In rural regions of southern Africa, subsistence farmers depend on livestock, charcoal, and small-scale agriculture to survive. These activities encroach on existing animal habitats and degrade their quality, pushing animals into croplands. Damage incurred from their visits is not trivial or reimbursed by the government. Some farmers report monthly damages of $30–80 from vervet monkeys alone, in countries where daily income averages less than $2.15. In that context, with few other options, killing a monkey becomes less of a crime than a last resort to defend their livelihoods.
Organizations like African Wildlife Foundation and Game Rangers International in Zambia work demonstrate that community education and alternative livelihood programs can effectively reduce retaliatory killing and dependence on hunting for meat or income.
- Alberts, S. C., Hollister-Smith, J. A., Mututua, R. S., Sayialel, S. N., Muruthi, P. M., Warutere, J. K., & Altmann, J. (2009). Seasonality and long-term change in a savanna environment. In D. K. Brockman & C. P. van Schaik (Eds.), Seasonality in primates: Studies of living and extinct human and non-human primates (pp. 157–196). Cambridge University Press. https://doi.org/10.1017/CBO9780511641953.007
- Alemneh, C., & Girma, Z. (2024). Diurnal Activity Budgets and Feeding Habits of Grivet Monkey (Chlorocebus aethiops aethiops) in Fragmented Moist Afromontane Forest. African Journal of Ecology, 62(4). https://doi.org/10.1111/aje.13339
- Bailey, A. L., Lauck, M., Ghai, R. R., Nelson, C. W., Heimbruch, K., Hughes, A. L., Goldberg, T. L., … International Vervet Research Consortium. (2016). Arteriviruses, pegiviruses, and lentiviruses are common among wild African monkeys. Journal of Virology, 90(20), 9434–9448. https://doi.org/10.1128/jvi.00573-16
- Borgeaud, C., Sosa, S., Bshary, R., Sueur, C., & van de Waal, E. (2016). Intergroup variation of social relationships in wild vervet monkeys: A dynamic network approach. Frontiers in Psychology, 7, 915. https://doi.org/10.3389/fpsyg.2016.00915
- Bourlière, F. (1985). Primate communities: Their structure and role in tropical ecosystems. International Journal of Primatology, 6(1), 1–26. https://doi.org/10.1007/BF02693694
- Clayton, C. L. (2025). Male and female contributions to sexual dimorphism (Master’s thesis). University of Arkansas. Retrieved from https://scholarworks.uark.edu/etd/5778
- Cramer, J. D. (2012). Scrotal color and signal content among South African vervet monkeys (Chlorocebus aethiops pygerythrus) (Doctoral dissertation). ProQuest Dissertations Publishing. https://www.proquest.com/
- Disotell, T. R. (1996). The phylogeny of Old World monkeys. Evolutionary Anthropology, 5(1), 18–24. https://doi.org/10.1002/(SICI)1520-6505(1996)5:1<18::AID-EVAN6>3.0.CO;2-S
- Dolotovskaya, S., Torroba Bordallo, J., Haus, T., Noll, A., Hofreiter, M., Zinner, D., & Roos, C. (2017). Comparing mitogenomic timetrees for two African savannah primate genera (Chlorocebus and Papio). Zoological Journal of the Linnean Society, 181(2), 471–483. https://doi.org/10.1093/zoolinnean/zlx001
- Dubuc, C., Nelson, E., Semel, B., Teichroeb, J. A., & Isbell, L. A. (2021). Be early or be tolerated: Vervet monkey (Chlorocebus pygerythrus) foraging strategies in a dispersed resource. Animal Behaviour, 176, 1–15. https://doi.org/10.1016/j.anbehav.2021.03.010
- Fairbairn, D. J., & Cox, R. M. (2024). Sexual dimorphism. In E. J. Eisen, K. R. Zinn, & J. R. Mason (Eds.), Encyclopedia of reproduction (2nd ed., Vol. 6). Elsevier. https://doi.org/10.1016/B978-0-443-15750-9.00008-2
- Freeman, N. J., Pasternak, G. M., Rubi, T. L., Barrett, L., & Henzi, S. P. (2012). Evidence for scent marking in vervet monkeys? Primates, 53, 311–315. https://doi.org/10.1007/s10329-012-0304-8
- Frost, P. G. H. (1996). The ecology of miombo woodlands. In B. Campbell (Ed.), The miombo in transition: Woodlands and welfare in Africa (pp. 11–57). Centre for International Forestry Research.
- Gagnon, C. M., Svardal, H., Jasinska, A. J., Cramer, J. D., Freimer, N. B., Grobler, J. P., Turner, T. R., & Schmitt, C. A. (2022). Evidence of selection in the uncoupling protein 1 gene region suggests local adaptation to solar irradiance in savannah monkeys (Chlorocebus spp.). Proceedings of the Royal Society B: Biological Sciences, 289(1982), 20221254. https://doi.org/10.1098/rspb.2022.1254
- Hart, D., Colquhoun, I. C., & Sussman, R. W. (Eds.). (2022). The natural history of primates: A systematic survey of ecology and behavior. Rowman & Littlefield Publishers.
- Haus, T., Akom, E., Agwanda, B., Hofreiter, M., Roos, C., & Zinner, D. (2013). Mitochondrial diversity and distribution of African green monkeys (Chlorocebus Gray, 1870). American Journal of Primatology, 75(4), 350–360. https://doi.org/10.1002/ajp.22113
- International Union for Conservation of Nature. (2019). The IUCN Red List of Threatened Species (Version 2019-3). Retrieved June 12, 2025, from https://www.iucnredlist.org
- International Union for Conservation of Nature (IUCN). (2022, December 6). West and Central Africa’s wildlife in trouble, shows new IUCN report. IUCN. Retrieved June 12, 2025, from https://www.iucn.org/content/west-and-central-africas-wildlife-trouble-shows-new-iucn-report
- Isbell, L. A., & Bidner, L. R. (2016). Vervet monkey (Chlorocebus pygerythrus) alarm calls to leopards (Panthera pardus) function as a predator deterrent. Behaviour, 153(5–6), 591–606. https://doi.org/10.1163/1568539X-00003371
- Kazaba, P. K. (2019). ‘Non-protected’ primates as bushmeat, pets and pests in southeastern Democratic Republic of Congo. Journal of Threatened Taxa, 11(3), 13251–13260. https://doi.org/10.11609/jott.4669.11.3.13251-13260
- Kingdon, J., Gippoliti, S., Butynski, T. M., & de Jong, Y. (2008). Chlorocebus pygerythrus. The IUCN Red List of Threatened Species 2008: e.T136271A4267738. https://doi.org/10.2305/IUCN.UK.2008.RLTS.T136271A4267738.en
- Magden, E. R., Mansfield, K. G., Simmons, J. H., & Abee, C. R. (2015). Nonhuman primates. In J. G. Fox, L. C. Anderson, G. M. Otto, K. R. Pritchett-Corning, & M. T. Whary (Eds.), Laboratory animal medicine (3rd ed., pp. 771–930). Academic Press. https://doi.org/10.1016/B978-0-12-409527-4.00017-1
- Mekonnen, A., Fashing, P. J., Bekele, A., Hernandez-Aguilar, R. A., Rueness, E. K., & Stenseth, N. C. (2018). Dietary flexibility of Bale monkeys (Chlorocebus djamdjamensis) in southern Ethiopia: Effects of habitat degradation and life in fragments. BMC Ecology, 18(4). https://doi.org/10.1186/s12898-018-0161-0
- Mittermeier, R. A., Rylands, A. B., & Wilson, D. E. (2013). Handbook of the mammals of the world: Volume 3 – Primates. Lynx Edicions.
- Nyirenda, V. R., Myburgh, W. J., Reilly, B. K., Phiri, A. I., & Chabwela, H. N. (2013). Wildlife crop damage valuation and conservation: Conflicting perceptions by local farmers in the Luangwa Valley, eastern Zambia. International Journal of Biodiversity and Conservation, 5(13), 793–804. https://doi.org/10.5897/IJBC2013.0609
- Richardson, M., & Jaffe, K. (2025). Chlorocebus cynosuros. In N. Rowe & M. Myers (Eds.), All the world’s primates. Retrieved June 30, 2025, from https://www.alltheworldsprimates.org
- Schad, L. (2023). Determinants of vocal usage in the genus Chlorocebus (Doctoral dissertation, University of Göttingen). https://doi.org/10.53846/goediss-10041
- Schapiro, S. J., & Jorgensen, M. (2017). Behavioral management of Chlorocebus spp. In S. J. Schapiro (Ed.), Handbook of primate behavioral management (1st ed., pp. 339–367). CRC Press. https://doi.org/10.1201/9781315120652-1
- Sudarikova, E. (2021). On the problem of craniological differentiation in two species of the green monkeys: Chlorocebus pygerythrus and C. aethiops (Cercopithecidae, Primates). Novitates Theriologicae, 12, 292–299. https://doi.org/10.53452/nt1246
- Takahashi, M. Q., Rothman, J. M., Raubenheimer, D., & Cords, M. (2019). Dietary generalists and nutritional specialists: Feeding strategies of adult female blue monkeys (Cercopithecus mitis) in the Kakamega Forest, Kenya. American Journal of Primatology, 81(9), e23016. https://doi.org/10.1002/ajp.23016
- Teutloff, N., Meller, P., Finckh, M., Cabalo, A. S., Ramiro, G. J., Neinhuis, C., & Lautenschläger, T. (2021). Hunting techniques and their harvest as indicators of mammal diversity and threat in Northern Angola. European Journal of Wildlife Research, 67(6). https://doi.org/10.1007/s10344-021-01532-5
- Turner, T. R., Schmitt, C. A., Cramer, J. D., Lorenz, J., Grobler, J. P., Jolly, C. J., & Freimer, N. B. (2018). Morphological variation in the genus Chlorocebus: Ecogeographic and anthropogenically mediated variation in body mass, postcranial morphology, and growth. American Journal of Physical Anthropology, 166(4), 682–707. https://doi.org/10.1002/ajpa.23459
- Turner, T. R., Schmitt, C. A., & Cramer, J. D. (2019). Savanna monkeys: The genus Chlorocebus. Cambridge University Press.
- UN Environment Programme. (2023, January 9). Researchers strive to end human–wildlife conflicts in Southern Africa. UNEP News and Stories. Retrieved June 12, 2025, from https://www.unep.org/news-and-stories/story/researchers-strive-end-human-wildlife-conflicts-southern-africa
- Wallis, J. (2013). Chlorocebus cynosuros, Malbrouck monkey. In R. A. Mittermeier, A. B. Rylands, & D. E. Wilson (Eds.), Handbook of the mammals of the world: Volume 3 – Primates. Lynx Edicions.
- Yitayih, Y., Ejigu, D., & Mola, M. (2023). Population size and human–grivet monkeys (Chlorocebus aethiops) conflict in Zegie peninsula, Bahir Dar, Ethiopia. Heliyon, 9(4), e15101. https://doi.org/10.1016/j.heliyon.2023.e15101
- Zinner, D., & Roos, C. (2016). Primate taxonomy and conservation. In M. T. Waller (Ed.), Ethnoprimatology (pp. 193–213). Springer. https://doi.org/10.1007/978-3-319-30469-4_11
Written by Alyssa Hanes, Jul 2025
