Brown Greater Galago, Otolemur crassicaudatus
BROWN GREATER GALAGO
Otolemur crassicaudatus

Geographic Distribution and Habitat
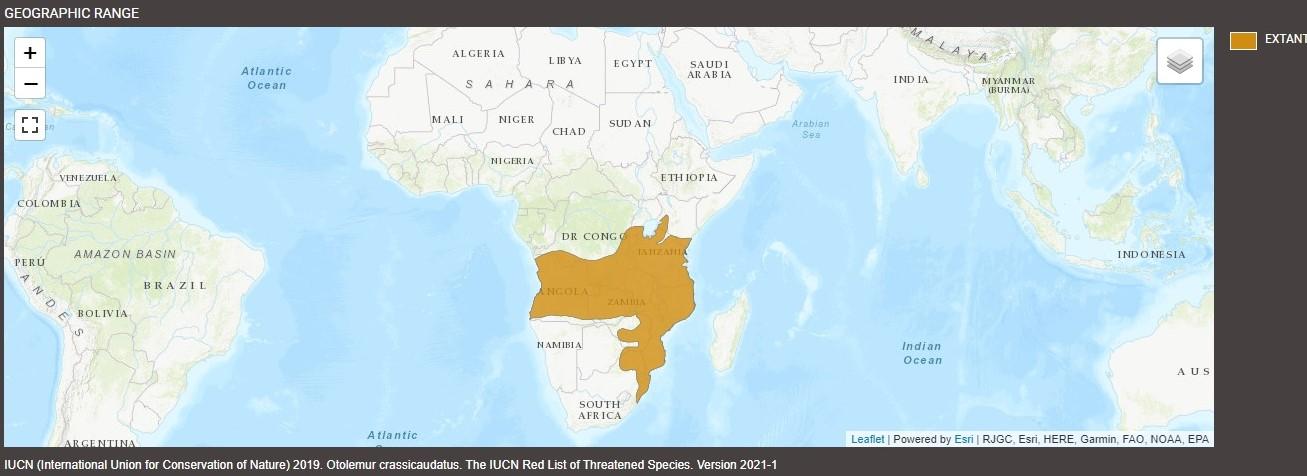
The brown greater galago (Otolemur crassicaudatus), also known as the thick-tailed greater galago, greater bushbaby, greater galago, large-eared greater galago, or thick-tailed bushbaby, is a prosimian species from southeastern Africa. They range widely across a large number of countries, such as Angola, Burundi, Congo, the Democratic Republic of the Eswatini, Kenya, Malawi, Mozambique, Rwanda, South Africa, Tanzania, Zambia, and Zimbabwe.
Brown greater galagos are primarily arboreal (tree-dwelling) and prefer to live in relatively wet, higher rainfall areas with high canopy cover, such as riverine forests, woodlands, and wet savannah with high gum tree composition. The altitude of their habitat can range from sea level up to 5,900 ft (1,800 meters).
The brown greater galago belongs to Otolemur genus in the Galagidae family. There are currently two recognized greater galago species within the Otolemur genus: the brown greater galago (Otolemur crassicaudatus), which is the largest of the galagos; and the slightly smaller Garnett’s greater galago (Otolemur garnettii), which has a more limited distribution on the coast of East Africa. There is some ambiguity surroundings the taxonomy of brown greater galago subspecies, with three or four recognized depending on the source. The International Union for Conservation of Nature, which is considered the authority in such matters, currently lists four subspecies:
- The northern silver galago, O. c. argentatus, endemic to Kenya, Rwanda, Tanzania, and Uganda
- The South African large-eared galago, O. c. crassicaudatus (the nominate species), endemic to the Mpumalanga, Limpopo Province, and KwaZulu-Natal provinces of South Africa
- The Tanganyika large-eared galago, O. c. kirkii, which ranges through Malawi, Mozambique, and Zimbabwe
- The Miombo silver galago, O. c. monteiri, found in Angola, Kenya, Malawi, Mozambique, Rwanda, and Zambia
As the questions of subspecies identity continue, more genomic studies with larger sample sizes may determine futher differences at the species and subspecies levels.
The brown greater galago is the largest of the galagos. The head-to-body length ranges from 10 to 15.8 in (25.5–40 cm), and tail length is 14–18 in (35–45 cm). Sexual dimorphism in body size exists, with males being heavier than females. On average, males can weigh up to 4.4 lbs (2 kg) and females weigh around 2.2 lbs (1 kg).
One lifespan record for the subspecies silvery greater galago indicates that they may live for up to 22.7 years in captivity. However, in the wild, individuals may reach 10 to 15 years of age.
Bright-eyed and bushy-tailed, the brown greater galago resembles a cat, especially when walking on all four limbs. Hindlimbs are slightly longer than forelimbs, and they primarily move by quadrupedally walking and running on all fours. If distressed, they can leap about 10 ft (3 meters) between trees and hop along the ground bipedally. Their hind feet consist of a large opposable big toe, which provides them with the ability to maintain a grip when hanging upside-down on a horizontal branch—which they often do to reach the gum source.
The face of the brown greater galago is dominated by large eyes and ears. Because of the relatively large size of the eyes, galagos have a limited range of eye movements and compensate for this with more head movements to explore novel objects. Galagos are able to localize insect prey with the help of their large and independently mobile ears, and frequently use this sense to detect prey that is out of sight. They can also search for insects visually and find sources of gum using their keen sense of smell.
The pelage of Tanganyika large-eared galago subspecies is paler than that of South African large-eared galagos, and the tail and extremities have a rufous brown tint. The silvery greater galago, as the name suggests, is a silvery-gray subspecies with dark hands and feet; their undersides are a creamy yellow hue. The tail is often nearly white. Pelage variation is likely linked to varying needs for camouflage in different vegetation types, and possibly different seasons.

The omnivorous diet of brown greater galagos consists of fruits, gum, seeds, and insects but varies considerably from season to season depending on the availability of each type of food source. Gum constitutes about 62% of the food and is eaten throughout the year, especially in winter, when other foods are in short supply. Fruit represents 21%, mostly in summer, and insects 5%. While feeding on gum, the galagos lick away for up to 20–30 minutes at a time and chew the bark to expose more gum. Sharing is caring, and one may find 2 to 3 galagos licking gum at the same spot, without fighting. While foraging, the galagos may rummage through the leaves on the ground to find fallen seed pods or fruits. They have never been observed drinking water directly from the water bodies and obtain their moisture requirements from food.
Brown greater galagos are nocturnal and spend the day sleeping in dense vegetation, on branches at a height of 16–40 ft (5–12 m) above the ground. They begin their nightly activity after sunset and return to their nests before sunrise. After waking up, they freshen up by grooming themselves or one another. The post wake-up grooming ritual is followed by direct movement to a food tree, where they spend up to two hours. When searching for sources of gum and insects, they often forage alone, but if large fruit trees are in season, they forage together. After feeding, they take a short nap or may partake in grooming activities. About 30–60 mins before dawn, with their bellies full, the galagos move to their favorite sleeping tree. Generally, a particular tree is used repeatedly as a sleeping site. While sleeping, group size can range from two to six individuals. If the group must move sleeping sites during the night, they emit a call to aid movement together as a group. Females with offspring put their engineering skills to use by building nests, which are platforms of broad-leaf twigs, woven together so that the family can rest comfortably, away from predators on the ground.
Galagos can effectively locate insect prey in darkness with the help of their large ridged ears, which amplify sound. Each ear can move independently to zero in on the prey’s location.
Galagos practice urine-washing, a behavior in which urine is wiped across the soles of both the feet, to reduce friction during movement and to leave scents for passersby.
Daily Life and Group Dynamics
Each group of brown greater galagos has a fixed home range, and densities of up to 324 animals per square mile (125 per km2) have been observed. There are several resting sites within a group’s home range. Males tend to sleep alone and, as they mature sexually, separate themselves from the group to gain personal space. The home range of a male may overlap with the home range of several females. They live in a social organization with multiple males and females overlapping in range use.
Most of the foraging is done on the ground. As a result, they may become victims of road accidents in areas where they occur in high densities. Territories are physically marked with bite marks, in addition to secretions from a gland on the chest, and with urine that is rubbed on hands and feet.
Communication
Brown greater galagos have a vast repertoire of calls, which vary according to the context and levels of arousal they face. In fact, one of their common names, bushbabies, is derived from their distinctive loud calls, which sound like the cry of a human baby. These cries are heard often during the breeding season but are also heard throughout the year and may be for marking territories.
In especially threatening circumstances, such as the presence of a potential predator, the adults give continuous calls consisting of several different sounds that may gradually increase in intensity and could last up to 1.5 hours. The calls start with a low-intensity nasal sound with the adults staring and pointing their noses towards the object of interest while sniffing. The sniff then progresses to a “knock” vocalization, to which up to three animals may join in on. Knocks are also common at the time of mating. An intermediate vocalization is called a “creak,” which may also occur during social interactions in addition to potential predator sightings. When a galago feels startled or threatened by sudden movements, he or she emits a loud call called “squawk,” which may turn into whistles when highly agitated. Sometimes during fights or during the mating season, adults scream at each other.
Toddlers have their own set of vocalizations, with the youngsters making a number of clicking, squeaking, or buzzing noises. Newborn infants emit squeaks while being carried by their mother. If the infant happens to lose physical contact with their mother, they emit clicks. If the youngster happens to get lost or cannot find their family, they will buzz and frantically search by moving branches.
In addition to vocal communication tactics, brown greater galagos practice urine-washing, a behavior in which urine is dribbled onto the palm of one hand and wiped across the soles of both the feet. Urine-washing enables galagos to leave a scent trail behind them as they pursue their nightly activities. Brown greater galagos also have a bare patch of skin on the chest, which is rich in scent glands and used for scent-marking. The major component of the chest gland secretion is benzyl cyanide, and the complex odor changes with time as the volatile components evaporate, enabling neighbors to track one another’s movements.
Reproduction and Family
Brown greater galagos attain sexual maturity between 10 and 18 months of age with the first conception occurring from 15 to 18 months. The timing of births and the start of nest-building activity coincide with the onset of the rainy season when food supplies increase in abundance and cover becomes denser. Therefore, different locations have different breeding seasons. Gestation lasts for approximately 132 days, and they usually give birth to twins or triplets. Infants may nurse for up to 5 months on fat and protein-rich milk, and wean off when they turn around 3 to 5 months of age. Reproductivity drastically drops at about 6 years of age, with the probability of getting pregnant past this age being low.
Before infants can move on their own, the females carry their infants in their mouths. In cases of triplets, one is carried in the mouth and two on the fur of the back when moving from one nest to the other. The mother tends to forage alone and leaves the kids at the resting site. At about 10–16 weeks, the infants are able to follow their mother around, play, and socialize. At 10 months, the infants leave their sleeping sites and start feeding independently.

The frugivorous dietary habits of the brown greater galago allow them to play a role in seed dispersal for indigenous plant species. As arboreal, fruit-eating primates, they can carry seeds over relatively large distances and perform a more scattered seed dispersal function compared to terrestrial seed-dispersers.


The brown greater galago is listed as Least Concern on the International Union for Conservation of Nature (IUCN, 2018) and Natural Resources Red List of threatened species, despite several populations being considered locally threatened due to habitat destruction and fragmentation. For example, previously, the greater galagos could be found commonly all-around Lake Victoria, but they have now nearly vanished from the area.
While the current habitats for the galagos are fragmented, the southern-most limits of the species are even more fragmented, due to reduced tree cover and the region being drier. These habitats are further fragmented by economic activities such as timber plantations, agriculture, livestock farming, and urban development. For example, in South Africa, Limpopo Province, pine and eucalyptus plantations and residential housing expansion reduced forest cover by 20% over a 16-year period between 1990 and 2006. Inability to migrate between different forests leads to inbreeding in isolated subpopulations. Other human activities such as illegal pet trade, traditional medicine, and poaching for human consumption also result in population declines.
The brown greater galago is listed in Appendix II of the Convention on International Trade in Endangered Species (CITES), an international agreement between governments whose goal is to ensure that international trade in specimens of wild animals and plants does not threaten their survival.
Especially in South Africa, key conservation areas such as wildlife game reserves and parks have been established. Kruger National Park holds strong populations. Ongoing research, for example at the African Primate initiative for Ecology and Speciation at the University of Fort Hare in South Africa, is aiming to characterize crucial habitat characteristics necessary for the survival and to assess which populations are most at risk of extinction throughout sub-Saharan Africa. Protected area expansion and connection of forest habitats through corridors should be developed to provide a safe migration route for these animals. Galagos adapt well and breed very successfully in captivity, and such facilities can be used to reintroduce this species into conservancies and other protected areas.
- Gursky, Sharon L. and Nekaris, K. A. I. (2007). A Comparison of Calling Patterns in Two Nocturnal Primates, Otolemur crassicaudatus and Galago moholi as a Guide to Predation Risk. Chapter 9, 206–221.
- Masters, J. and Bearder, S. (2019). Otolemur crassicaudatus. The IUCN Red List of Threatened Species 2019.
- Masters, J. and Génin F. (2016). A conservation assessment of Otolemur crassicaudatus. The Red List of Mammals of South Africa, Swaziland, and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
- Nash, L. T., Bearder, S. K., and Olson, Todd R. (1989). Synopsis of Galago species characteristics. 10(1), 57–80.
- Eaglen, Robert H. and Simons, Elwyn L. (1980). Notes on the Breeding Biology of Thick-Tailed and Silvery Galagos in Captivity. Journal of Mammalogy, 61(3), 534–537.
- Hendrickx, Andrew G. (1995). Nonhuman Primates in Biomedical Research || Reproductive Biology, 147–191.
- Prince Rainier III HRH, Bourne GH (1977). Primate conservation. New York: Academic Press.
Written by Divya Pawar, June 2022
