Black Bearded Saki, Chiropotes satanas
BLACK BEARDED SAKI
Chiropotes satanas

Geographic Distribution and Habitat
The black bearded saki, also known as the bearded saki, black cuxiú, and black saki, is one of five species of bearded saki monkey that reside within the Amazon rainforest. In Brazil, people affectionately refer to these monkeys as cuxiú, a name that indigenous communities have long used. Some researchers suggest using the common name cuxiú for bearded sakis to avoid confusion with other monkeys also called sakis (in the genus Pithecia).
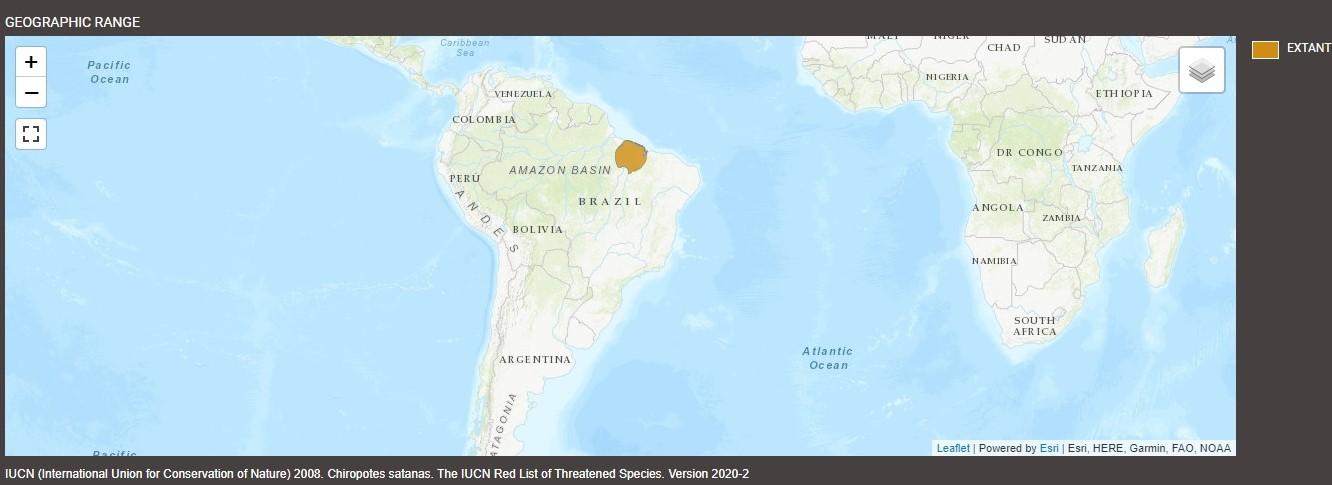
Black bearded sakis are found in a small region of Brazil’s eastern Amazonia, located east of the Tocantins River, extending to the Grajaú River. They tend to inhabit primary tropical rainforests, where dense greenery provides cover from predators. Otherwise, they inhabit the Cerrado, South America’s tropical savanna, coastal mangrove rainforests, and terra firma, or dry-land, forests. They occupy elevations ranging between 49 and 1300 feet (15-400 m) above sea level. In the Brazilian states of Pará and part of Maranhão, where black bearded sakis live, the climate follows a humid equatorial cycle with a marked wet season from December to June and a distinct dry season from July to November. Annual rainfall in their habitat typically ranges from 40 to 60 inches (102-152 cm). Temperatures generally stay within a narrow range from 77 to 82 ºF (25 to 28 ºC) year-round, typical of the tropical equatorial climate.
Seasonal changes, especially shifts in rainfall, have multiple effects on how black bearded sakis interact with their surroundings. During the wet season, many trees produce large, unripe fruits with hard husks—exactly what sakis are adapted to exploit with their powerful jaws. As a result, they tend to spend more time feeding on abundant seeds, travel more directly between productive trees, and show relatively stable daily routines due to more predictable sources of food. In the dry season, fruit production often declines or shifts in species composition. Sakis respond by broadening their diet to include more ripe fruits, flowers, or insects, increasing their foraging time and effort, and visiting a wider range of plant species. In times of high fruit abundance, groups may maintain smaller, more stable home ranges where there are many high-value food items. During periods when fruits are not plentiful, they often expand their daily travel distances, ranging more widely to locate sufficient food. Their movement is typically fast and coordinated, but seasonal food scarcity can make routes less predictable as they search for patchy resources. Black bearded sakis live in relatively large, cohesive groups. Seasonal food changes can subtly alter social behavior: when food is plentiful, group cohesion is strong, with many individuals feeding in the same trees; when resources are limited, groups may spread out more while foraging to reduce competition, even if they remain socially bonded overall. Finally, seasonal variation in rainfall and daylight can influence daily activity. In wetter periods, sakis may adjust their activities to avoid heavy rain, sometimes showing brief pauses in movement. Conversely, in drier periods, longer foraging times may extend active hours slightly, especially if food is harder to find.
Size, Weight, and Lifespan
Male black bearded sakis are slightly larger than females, averaging 15 to 16.5 inches (38.1-42 cm) in head-body length and weighing between 5.5 and 8.8 pounds (2.5-4.0 kg). Females average 13.4 to 15.4 inches (34-39 cm) in head-body length, and typically weigh between 4.4 and 7.7 pounds (2.0-3.5 kg). Tail length does not differ very much between sexes; males range between 14.2 and 16.5 inches (36-42 cm), while those of females range between 14.2 and 16.1 inches (36-41 cm). These differences in size and weight are examples of sexual dimorphism or noticeable physical distinctions between the sexes.
The life expectancy of bearded sakis is approximately 15 years in the wild.
Appearance
Although predominantly black in color, some black bearded sakis have a lovely brownish hue across their shoulders and upper back. Their fur is dense and covers the entire body in a thick mat. Both males and females sport a thick beard on the chin and puffy, rounded fur, supported by bulging, tennis ball-like muscles on top of their heads, called coronal tufts. These two traits combine to give the black bearded saki a head with an endearing heart-shaped appearance. There is prominent sexual dimorphism concerning these two charismatic traits: the beards of males are longer and thicker than those of females, and the characteristic bulging forehead is less pronounced in females when compared to that of males.
Hairless faces with round, dark eyes give them an expressive and curious look. A feature of all monkeys found in the Americas, black bearded sakis have a dry, platyrrhine nose with nostrils set widely apart; in fact, platyrrhine means flat-nosed in Greek. Now, what’s the actual significance of this type of nose? Platyrrhine monkeys are almost exclusively arboreal, and a broad, flat nose with nostrils directed outward or sideways is highly efficient for maneuvering in the dense, high-humidity canopy of the tropical rainforest. Now you nose—I mean, know!
Black bearded sakis have a slender, well-defined body shape, often described as robust yet lean. This body type makes them highly agile, aiding their long-distance locomotion within the high canopy of their forested habitat. Black bearded sakis also have shorter forelimbs than hind limbs, and small, not fully opposable thumbs—features that are perfect for scurrying through the canopy.
Black bearded sakis lack fully prehensile tails, meaning they do not use their tails to grasp branches or bear their weight. However, they can use them as a balance when moving through trees, or as extra support when stretching out for hard-to-reach fruits.
A less visible but still significant trait is their uniquely powerful jaws and specialized molars. Their canine teeth are wedge-shaped, allowing them to tear through the tough skins of unripe fruits, and their molars are flat, designed to crack open seeds. These are highly effective adaptations for chewing through thick, tough husks of unripe fruit to access nutrient-rich seeds inside. No fruit is safe from these adorable monkeys!

Diet
Black bearded sakis are primarily frugivores, meaning they mostly eat fruits. Black bearded sakis are also granivores, or seed-eaters, favoring those of the juicy fruits that make up the majority of their diet. During the dry season, when fruits are less abundant, they also eat leaves and insects such as caterpillars, termites, and gall wasps. Insects are scooped up easily with their agile fingers, and their excellent eyesight makes them easy to find. Termites, caterpillars, and gall wasps are all delicious meals for black bearded sakis. They will also lie down near ant mounds and allow the ants to crawl into the fur of their arms. Once they’ve collected enough ants to make a snack, they can lie back and enjoy easy pickings, plucking them out of their fur one by one.
Black bearded sakis prefer to eat seeds from unripe fruit, allowing them to remain in the canopy and stay comfortable. However, chewing through unripe fruit to access the seeds inside can be a difficult task. As mentioned in the Appearance section, black bearded sakis have developed very strong jaws with teeth specifically designed to access and consume seeds. They tend to choose unripe fruit because they are a readily available source of food, and they can eat it without competing with other fruit-eating primates. Despite having eyesight keen enough to detect tiny insects, like many Amazonian monkeys, black bearded sakis have dichromatic vision and cannot see the color red. Without this ability, it is very difficult for them to assess a fruit’s ripeness by color.
In what scientists call geophagy, black bearded sakis occasionally consume soil or clay, although this is rare. Research suggests that consuming certain minerals may help black bearded sakis bind and process harmful toxins found in the seeds they eat.
Behavior and Lifestyle
Black bearded sakis are arboreal, meaning they spend their time high in the trees. They tend to move quadrupedally, meaning they scramble on the tops of branches using all four limbs. They don’t jump between trees or tree limbs unless it’s necessary, and instead, prefer to walk between trees with branches close together. Nevertheless, they can jump and land on all fours.
They are diurnal, meaning they are active during the daylight hours, and spend that time searching for food, resting, and interacting with other group members. Studies of the daily activity budgets of black bearded sakis suggest they spend 58.5% of their time traveling, 19.8% dedicated to foraging, 13.8% spent resting, and 7.9% reserved for other activities, likely grooming and playing.
Besides humans, predators of bearded sakis include boa constrictors, jaguars, ocelots, and raptors, specifically the harpy eagle. To avoid predators, they stay high in the trees and use their agility to escape from potential threats. While remaining high up in the trees helps against ground-based predators, black bearded saki tend to inhabit areas with thick foliage, providing ample cover from watchful predators, especially aerial ones.
Black bearded sakis do not construct sleeping nests. Rather, they choose the tops or crowns of the tallest trees with plenty of foliage to protect them from predators. Groups may use the same sleeping site on consecutive nights, and large groups distribute themselves among several trees. Curling the tail around the body, black bearded sakis huddle against one another in the branches to sleep the night away comfortably.
Dinner and a show! Adapted for an agile life in the trees, the light frame of black bearded sakis allows them to suspend themselves from branches by their hind limbs while feeding.
Primate playdate! Young black bearded sakis play with juvenile howler monkeys and capuchins in the wild.
The home range of a black bearded saki group ranges between 42-245 acres (17-99 ha), and their daily traveling range varies between 1.2-3.9 miles (1.9-6.3 km).
The social life of these adorable little primates is quite fascinating, although extensive scientific knowledge remains limited. They live in large social groups of around 40 members with males and females of various ages. The members of this larger group will break into smaller groups while foraging and feeding, a social dynamic called fission-fusion. While the smaller groups may spend several days on their own, ultimately, they rejoin the larger group. Adult males frequently sit near and groom each other, rarely interacting with females or juveniles. Likewise, females spend time grooming juveniles and other females. Although they reach maturity at around 3 years of age, scientists are unsure whether males or females leave the group.
Occasionally, when traveling through the forest canopy, a group member may be left behind. Sakis are vulnerable when alone and may temporarily join troops of other primates until they can return to their birth group. Spider monkeys, squirrel monkeys, howler monkeys, and capuchins will temporarily tolerate sakis traveling, feeding, and resting amongst group members, and respond to the alarm calls of their temporary group mates (specifically, black-capped capuchin, the Guinean squirrel monkey, the Ka’apor capuchin, the red-handed howler monkey, and the black-handed tamarin).
Black bearded sakis use both vocalizations and body language to communicate.
Sound is a convenient way to keep in touch when dense foliage often prevents monkeys from keeping an eye on one another. By making high-pitched whistles throughout the day, members remain in regular contact with their fellow troop members.
While feeding, black bearded sakis sometimes make subtle chirping sounds that seem to convey a sense of satisfaction and were particularly frequent when adults were eating caterpillars. If an individual is suddenly disturbed, however, they emit a shrill cry. Purring is an important aspect of black bearded saki mating rituals. Females purr before mating, and males purr during the mating process. Infants also purr while nursing, and it strengthens a mother’s bond with her baby. Alarm calls are sharp, shrill cries that alert the group to danger, a predator, or a disturbance.
Black bearded sakis express excitement by shaking their bushy, fox-like tails. They may also wave their tails silently like a flag to make contact with group members.
Allogrooming or mutual grooming creates and strengthens social bonds, and is most frequent among males, although females and juveniles also groom each other.
Finally, concerning scent marking, adults of both genders have been observed rubbing their rumps back and forth repeatedly along branches. Males also wash their hands and feet with urine and wipe them on tree branches. These scent markers support social cohesion, help navigation, convey reproductive status to potential mates, and indicate social status and dominance. Researchers debate whether scent marking defines territorial boundaries for black bearded sakis.
Even with this available knowledge, the full complexities of black bearded saki communication methods are not yet well understood.
Little information is available about the reproductive habits of black bearded sakis. Although they live in groups with multiple males and females, some observations suggest that individuals may form temporary pair associations. When sexually receptive, females may present their rumps to males, lying in front of them with the tail raised. Before mating, males may sniff or lick her genitalia.
After a pregnancy period of approximately four to five months, females give birth during the rainy season to a single infant; this allows them to care for infants when trees are most likely to have abundant fruit. After three months, infants are weaned off milk and can move independently with the troop, foraging on their own. These life history estimates are based on a captive saki hybrid and may not be entirely accurate for wild black bearded sakis. What is known is that females give birth to a single infant every two years, to whom they are the primary caregiver until weaning. She will nurse, carry, and teach the infant how to survive until they can move with the troop independently. Black bearded sakis reach sexual maturity at around four years of age, but their dispersal habits once mature are poorly understood.
Infant bearded sakis have been reported to use their tails to help them cling to their mothers during early development.

As much of their diet consists of fruit, black bearded sakis aid in the regeneration of their forest habitat by dispersing seeds through their feces during their daily travels. As insectivores, black bearded sakis also serve as pest controllers through their consumption of insects and their larvae. Furthermore, as granivores that can consume seeds from unripe fruit, black bearded sakis have a food source available to them long before other seed predators can access it, further assisting in seed dispersal. Supplementing their diet with insects and leaves helps them survive when food is scarce. Their primary competitors for unripe seeds aren’t other primates, but birds with large beaks capable of cracking and eating large shells and seeds, such as parrots and macaws.
Finally, as prey, they help maintain local predator populations in their habitat, such as jaguars and harpy eagles.


The International Union for Conservation of Nature lists the black bearded saki as Endangered (IUCN, 2020), appearing on the IUCN Red List of Threatened Species.
Black bearded sakis are considered extinct in much of their original habitat, an area of the Brazilian Amazon that has been subject to decades of colonization and deforestation. They have the smallest range of any species within their genus, Chiropotes. Their populations are decreasing at alarming rates, with the number of mature individuals possibly lower than 2,500.
The largest threat to black bearded sakis is the same that threatens many endangered primates around the world: habitat loss and fragmentation. As human populations continue to encroach upon the vast Amazon, large swaths of rainforest are cleared to make way for cattle grazing and ranching, to provide wood for the lumber industry, and to establish highways and secondary roads. The addition of the Tucuruí hydroelectric dam has contributed to further habitat loss in their already limited range.
One of the last areas of forest where black bearded sakis are known to live is the Gurupi Biological Reserve. The 1,319 square mile (341,650 ha) protected federal reserve is under constant threat of destruction from local ranchers, timber companies, and illegal land settlers. More recently, the marijuana trade has razed large swaths of land for plantations. Researchers estimate that one-third of its original extent no longer exists.
Black bearded sakis aren’t great leapers, and habitat fragmentation leaves them vulnerable to predators. It also limits their capacity to make treks to known foraging sites, thereby decreasing the amount of food they have available. Descending from the canopy increases their vulnerability to predators, including humans, who hunt them for their meat and fur. Habitat fragmentation is likely increasing hunting pressure on the black bearded saki by providing easier access for human hunters, and by the loss of other preferred food sources.
The black bearded saki is listed in Appendix II of the Convention on International Trade in Endangered Species (CITES), an international agreement between governments whose goal is to ensure that international trade in specimens of wild animals and plants does not threaten their survival.
Black bearded sakis, as well as many other primate species, will go extinct if human activities continue unaltered; deforestation and human encroachment continue to limit the amount of suitable habitat for these beautiful animals.
Conservation actions required to save the black bearded saki include surveying remnant populations of the species, developing environmental education programs to alert local communities to the status of this species, and more effective management in protected areas such as the Gurupi Biological Reserve. Within the reserve, there is a need for improved policing against hunting and logging, as well as improved physical infrastructure, and intensifying surveillance and research activities. Finally, more research and monitoring efforts are urgently needed, especially regarding population trends. If this endearing primate of the Amazon is to stand a chance at surviving the ongoing plight of extinction, much will need to change.
- https://www.alltheworldsprimates.org/Members/Home/MasterPrimate.aspx?tid=255
- https://en.wikipedia.org/wiki/Bearded_saki
- https://en.wikipedia.org/wiki/Black_bearded_saki
- https://en.wikipedia.org/wiki/Cerrado
- https://www.facebook.com/groups/2489985901214334/posts/2621602971385959/
- https://grokipedia.com/page/black_bearded_saki
- https://www.iucnredlist.org/species/39956/10297662
- https://www.knowyourmammals.com/mammal-identification/black-bearded-saki-chiropotes-satanas/
- https://www.personal.kent.edu/~mnorconk/pdfs/Walker_Suzanne_E.pdf
- https://news.mongabay.com/2024/08/in-brazils-amazon-land-invasions-and-fires-threaten-a-protected-reserve/
- https://primate.wisc.edy/primate-info-net/pin-factsheets/bearded-saki/
- https://www.researchgate.net/publication/234126234_Occurrence_and_diet_of_the_black_bearded_saki_Chiropotes_satanas_satanas_in_the_fragmented_landscape_of_western_Maranhao_Brazil
Written by Sienna Weinstein, April 2026
