Black-Footed Gray Langur, Semnopithecus hypoleucos
BLACK-FOOTED GRAY LANGUR
Semnopithecus hypoleucos

Geographic Distribution and Habitat
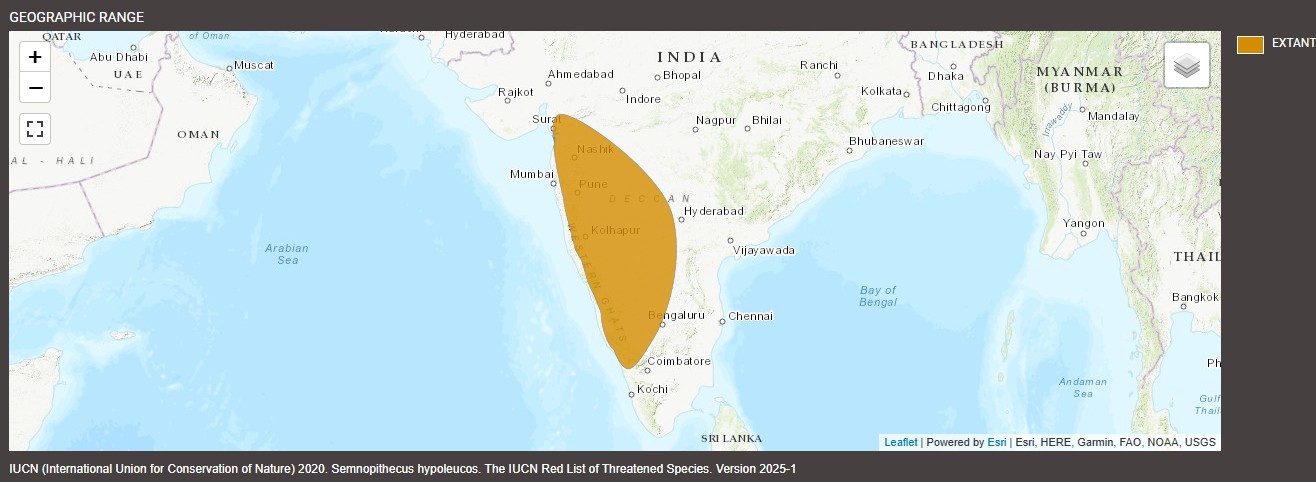
Black-footed gray langurs (Semnopithecus hypoleucos), also called dark-legged Malabar langurs or Malabar sacred langurs, are monkeys native to India. They are distributed across many territories, including Goa, Karnataka, Kerala, Andhra Pradesh, and Maharashtra. Found in a variety of ecosystems, they live at elevations between 328 to 3,937 feet (100 to 1,200 meters). Their ecosystems include tropical rainforests, moist deciduous forests, sacred groves, riparian forests, etc.
Because they are distributed over a large portion of the country, black-footed gray langurs have many geographic borders, including the Tapti River, Krishna River, and Indian Ocean. To the west, their habitats are defined by the Western Ghats, a mountain range that stretches along the western coast of the Indian peninsula. Stretching nearly 100,000 miles (16,000 kilometers), the Western Ghats is also known as the Sahyadri.
India has four distinct seasons throughout the year: winter (December to February), summer (March to May), monsoon (June to September), and post-monsoon, or autumn (October to November). Monsoon season is the primary rainy season in India. During this time, large parts of India’s southern and northwestern territory receive over 50% to 75% of their annual rainfall. Although it varies by region, between 7.87 and 11.81 inches (20 and 30 centimeters) of rainfall is collected over the course of these three months.
In western India, the average temperatures between summer and winter fluctuate significantly. In the summer, temperatures vary between 90 to 104 degrees Fahrenheit (32 to 40 degrees Celsius). Comparatively, in the winter, temperatures range between 50 to 59 degrees Fahrenheit (10 to 15 degrees Celsius).
Before modern taxonomic studies, black-footed gray langurs (Semnopithecus hypoleucos) were considered a subspecies of the northern plains gray langur (S. entellus). In 2003, research by Dr. Douglas Brandon-Jones suggested that black-footed gray langurs were part of the southern plains gray langur (S. dussumieri) population. Later studies, however, clarified that this population represents a distinct lineage, and some earlier hypotheses had proposed hybrid origins between the Nilgiri langur (Trachypithecus johnii) and southern plains gray langurs, though this is now largely discounted.
By 2014, comprehensive morphological and genetic studies recognized black-footed gray langurs as a distinct species, with several subspecies described based on geographic variation.
Size, Weight, and Lifespan
Black-footed gray langurs are sexually dimorphic, meaning males and females differ noticeably in size and build. Males are larger, with a head-to-body length of about 29 inches (74 cm) and a weight between 35 and 40 pounds (16–18 kg). Their tails are long, typically around 30–36 inches (76–91 cm), which helps with balance during arboreal movement. Females are smaller, averaging 25 inches (64 cm) in head-to-body length and weighing approximately 19 pounds (8.6 kg), with proportionally long tails of similar length to males.
In the wild, they can live up to 18 years.
Appearance
Black-footed gray langurs are long-limbed monkeys with several distinctive features. Their bodies are covered in long, silky fur, except for the face, hands, and feet. The fur varies in shades of cream, white, and gray, sometimes with a hint or patch of orange. Their triangular faces are framed by a tufted “crown” or corona of pale cream-colored hair.
The bare skin on their feet is naturally dark, giving rise to the name black-footed gray langur. Their hands and faces share this dark coloration. Because these areas have little or no fur, other facial features stand out clearly—such as their slender noses, vertical nostrils, and wide-set brown eyes.
They have a strong, hook-like grip that allows them to grasp rough branches securely, aided by their long fingers. As an evolutionary adaptation to their largely arboreal lifestyle, their thumbs are reduced in size, making their hands more efficient for swinging and climbing. Their tails are non-prehensile, meaning they cannot grasp or manipulate objects. Instead, the tail serves as a counterbalance, helping maintain stability while moving through the trees. For instance, when a langur crosses an unstable branch high in the canopy, it shifts its tail to counteract the body’s motion, maintaining balance and reducing the risk of a fall.

Diet
Gray langurs are primarily folivorous, meaning that most of their diet consists of leaves, both young and mature. However, they tend to favor young leaves, which are softer, more tender, and easier to digest than older, tougher foliage. Younger leaves also contain higher levels of nutrients and lower concentrations of defensive compounds like tannins, making them a more nutritious food source.
Langurs have multi-chambered sacculated stomachs that help them digest tough plant matter. Like ruminant species, their stomachs contain bacteria that ferment and break down fibrous plant materials, like cellulose. Cellulose is the main structural component of plant cells; it provides plants with the strength and rigidity they need to create cell walls. Once cellulose is broken down, the bacteria are neutralized, allowing leaf-eating monkeys to absorb necessary nutrients from the plant. This digestive characteristic is shared among all leaf-eating monkeys, which is why many appear to have rounded bellies after eating. Long rest periods provide them with enough time to properly digest their food and fuel the energy to take on the day!
In addition to leaves, black-footed gray langurs also consume other food products, such as fruits, flowers, seeds, bark, roots, etc. Plant species include mango (Mangifera indica), jamun (Syzygium cumini), guava (Psidium guajava), ber (Ziziphus mauritiana), karondas, also called caranadas cherries (Carissa carandas), and figs (Ficus carica).
Because India does not have a single fruiting season, different fruit species reach their peak abundance at various times of the year. For example, mangoes are most plentiful in summer, while guavas are more abundant during the monsoon season.
Although they also drink water from ground sources or tree hollows, most of their water is obtained through the moisture in their food, specifically from leaves, fruits, flowers, etc.
Behavior and Lifestyle
The habitats of black-footed gray langurs strongly shape their behavior and daily routines. They are both arboreal and terrestrial, meaning they divide their time between the trees and the ground. Among Asia’s leaf monkeys, gray langurs are the most terrestrial, spending more time on the ground than most other colobines.
They move primarily on all fours—a form of locomotion known as quadrupedalism—and are capable climbers, leapers, and runners. On occasion, they also perform short bipedal hops, propelling themselves with their hind legs, especially when crossing open ground or obstacles.
Black-footed gray langurs are diurnal, meaning they are active during the day and rest at night. They prefer sleeping sites at high elevations, typically in tall trees with broad, sturdy branches. Before settling, langurs appear to assess the size and stability of the tree to minimize the risk of falling while asleep.
In areas where natural trees are scarce or fragmented, they may instead choose man-made structures such as towers, rooftops, or electrical poles for sleeping. The specific type of sleeping site varies depending on the habitat and the level of human presence in the area.
Throughout their range, major threats to this species include: leopards (Panthera pardus), dholes (Cuon alpinus), tigers (Panthera tigris), wolves (Canis lupus pallipes), jackals (Canis aureus), indian rock pythons (Python molurus molurus), crested serpent eagles (Spilornis cheela), mountain hawk-eagles (Nisaetus nipalensis), and domestic dogs.
In their natural habitats, black-footed gray langurs spend much of their time on the ground, making them among the most terrestrial of the Asian leaf monkeys. In addition to foraging for ground vegetation, one reason for their time on the forest floor is to seek out minerals.
Through a behavior known as geophagy, langurs lick or consume soil, clay, or mud to obtain essential minerals such as sodium, calcium, and zinc. These minerals are believed to aid digestion by neutralizing plant toxins and acidity that result from their predominantly folivorous diet. What a rocking fact!
Black-footed gray langur troops vary in size, typically ranging from about 5 to 36 individuals. Males usually make up a smaller portion of the group, while females, juveniles, and subadults form the majority. Although group dynamics differ among troops, black-footed gray langurs generally follow a polygynous mating system.
In this system, a single dominant male mates with multiple females and serves as the primary breeder within the troop. Adult males attain leadership through displays of dominance and competition with rivals. Once in control, the male’s main role is to defend the group from predators and from other males attempting to take over.
As part of this competitive reproductive strategy, newly dominant males may commit infanticide—killing infants sired by the previous leader. The death of a mother’s infant triggers the return of her fertility sooner, enabling the new male to sire his own offspring. In males, reproductive success depends on both dominance and access to fertile females, and infanticide can be a means of securing that success and establishing a genetic legacy.
Male and female hierarchies are largely independent. While males compete primarily for breeding opportunities, females maintain their own social rankings, typically based on age, experience, physical condition, and matrilineal ties. Older females—especially those from high-ranking maternal lines—tend to hold higher status than younger or lower-ranking females.
Body language is an important means of communication for black-footed gray langurs. Individuals of all ages use postures and gestures to express affection, frustration, aggression, and sexual interest. For example, females in estrus signal their reproductive state by head-shaking and presenting the anogenital region, which may appear slightly swollen. Juvenile males often engage in play-mounting, a behavior that helps them practice adult social and sexual behaviors. Other forms of tactile communication include embracing, grooming, and even lip contact or “kissing.”
Vocalizations also play a key role in langur communication. Adult males produce loud calls, grunts, and barks to alert or assemble troop members, or to assert dominance and defend territory. Females are generally quieter but may squeak or grunt softly in response to a male’s call or during social interactions. Infants and juveniles emit high-pitched whines, shrieks, and cries when distressed, frightened, or separated from their mothers.
Estrus is the recurring phase in a female mammal’s reproductive cycle when she is sexually receptive and capable of conceiving. Commonly known as being “in heat,” a female langur in estrus often approaches a male and presents her anogenital region and hips as a signal of readiness to mate. The male then determines whether to accept or ignore her solicitation. Once conception occurs, the gestation period lasts about 200 days, or roughly 6 to 7 months—the time between fertilization and birth.
Infants are born weighing around 1 pound (0.46 kilograms) and are dependent on their mothers for many months. They cling tightly to their mother’s abdomen as she moves, even while leaping between trees. This behavior ensures the infant’s safety and provides early exposure to the physical and social challenges of life in the wild. In most cases, mothers are solely responsible for infant care, as adult males and juveniles do not participate directly in rearing.
Langur mothers also engage in alloparenting, a behavior in which other females help care for each other’s offspring. This cooperative system allows mothers to forage more efficiently and helps strengthen social bonds within the troop. Allomothers carry, groom, and protect infants, reducing the biological mother’s stress and providing her with brief opportunities to rest or feed. This shared care continues until infants become more independent and reach sexual maturity. Female gray langurs typically mature around 3 years of age, while males reach maturity around 5 years.
When males reach sexual maturity, they disperse from their natal troop to join or form bachelor groups—social units composed of young, non-breeding males. Life in a bachelor troop offers males opportunities to develop strength, practice dominance behaviors, and learn social strategies essential for future troop leadership. Eventually, mature males leave these groups to compete for dominance and attempt to take over a mixed-sex troop of their own.

Due to their dietary habits, black-footed gray langurs play an important role in forest regeneration. By feeding on leaves, they help open up the forest canopy, creating gaps that allow sunlight to reach the forest floor and promote the growth of new plants. In addition, langurs act as seed dispersers, maintaining plant diversity and supporting forest health. After consuming fruits, they digest and excrete seeds, or sometimes drop them, allowing seeds to escape competition and predation near the parent tree. By moving seeds to new locations, langurs facilitate the growth of new trees and enhance overall forest regeneration. Because of these combined contributions, langurs are often referred to as “forest gardeners”—and rightly so!


The International Union for Conservation of Nature lists black-footed gray langurs as Least Concern (IUCN, 2020), appearing on the IUCN Red List of Threatened Species.
This classification is largely due to the species’ relatively wide distribution and the absence of major widespread threats. However, the species does face conservation challenges. The primary threats include habitat loss and degradation, driven by agricultural expansion, human settlements, and habitat fragmentation. Secondary threats include deliberate fires, local hunting for food, and capture for the pet trade or traditional medicine, though these are generally less significant.
The black-footed gray langur is listed in Appendix I of the Convention on International Trade in Endangered Species (CITES), an international agreement between governments whose goal is to ensure that international trade in specimens of wild animals and plants does not threaten their survival.
Throughout India, they occur and are protected in Bhadra Sanctuary, Brahmagiri Sanctuary, Kudremukh National Park, Nagarahole National Park, Pushpagiri Sanctuary, Sharavathi Valley Sanctuary, Aralam Sanctuary, and Wayanad Sanctuary. They also occur in all protected areas of the Western Ghats and deacon plateau of Karnataka and Maharashtra.
To further ensure the longevity of this species, conservation efforts are needed and appreciated by scientists, researchers, ecologists, primatologists, etc. Research on taxonomy (on captive animals), life history, survey, ecology and behavior are especially needed. Further areas of research include habitat management, monitoring, and Population and Habitat Visibility Assessment.
- E. Huchard, P.M. Kappeler, N. Smit, C. Fichtel, & D. Lukas, The evolution of male–female dominance relations in primate societies, Proc. Natl. Acad. Sci. U.S.A. 122 (28) e2500405122, https://doi.org/10.1073/pnas.2500405122 (2025).
- Lu A, Sirimanna DGR, Wijayathunga L, Vandercone R, Salmi R. Mixed-species associations and attempted mating suggest hybridization between purple-faced and tufted gray langurs of Sri Lanka. Primates. 2021 Jan;62(1):11-17. doi: 10.1007/s10329-020-00852-z. Epub 2020 Aug 17. PMID: 32804328; PMCID: PMC7430210.
- Shield S, Jericevich R, Patel A, Jusufi A. Tails, Flails, and Sails: How Appendages Improve Terrestrial Maneuverability by Improving Stability. Integr Comp Biol. 2021 Sep 8;61(2):506-520. doi: 10.1093/icb/icab108. Erratum in: Integr Comp Biol. 2022 Oct 29;62(4):1145. doi: 10.1093/icb/icab198. PMID: 34050735; PMCID: PMC8633431.
- https://www.iucnredlist.org/species/167543916/17942313
- https://www.zoochat.com/community/media/black-footed-grey-langur-semnopithecus-hypoleucos.352220/
- https://en.wikipedia.org/wiki/Climate_of_India#:~:text=Traditionally%2C%20North%20Indians%20note%20six,twelve%20months%20into%20six%20parts.
- https://www.cpc.ncep.noaa.gov/products/assessments/assess_96/india.html#:~:text=Indian%20summer%20monsoon,58b
- https://primate.wisc.edu/primate-info-net/pin-factsheets/pin-factsheet-gray-langur/
- https://en.wikipedia.org/wiki/Black-footed_gray_langur
- https://zooatlanta.org/amazing-adaptations-of-the-angolan-colobus/
- https://www.taprobanica.org/~file/5_trachypithecusgeei_roynagarajan_taprobanica_8_1_33_36-e31fe-3349_762.pdf?b213411–#:~:text=Sleeping%20tree%20characteristics:%20The%20langurs,minimize%20the%20danger%20of%20falling.
- https://www.veterinarypaper.com/pdf/2025/vol10issue3/PartB/10-3-10-737.pdf
- https://link.springer.com/rwe/10.1007/978-3-030-85493-5_1996-1#:~:text=Definition,to%20prevent%20these%20violent%20acts.
- https://www.bioexplorer.net/animals/mammals/monkeys/black-footed-gray-langur/
- https://www.borneonaturefoundation.org/wildlife/squeaks-cries-and-whines-sounds-of-the-red-langur-monkey/
- https://zoosprint.org/index.php/zp/article/view/2833/2635
- https://www.sciencedirect.com/science/article/pii/S1146609X19302784#:~:text=Frugivorous%20animals%20contribute%20differently%20to,Chapman%20and%20Onderdonk%2C%201998).
- https://www.researchgate.net/publication/329109051_The_function_of_geophagy_in_Nepal_gray_langurs_Sodium_acquisition_rather_than_detoxification_or_prevention_of_acidosis?__cf_chl_tk=EX8drCPEtxHZ3u1xs0zKfGP1EkK9bC9henR3vHcay_M-1761238551-1.0.1.1-KWntSN8BvkPrl9A0mgltYcBYKlJEpKbesj2JP0_rqSg
Written by Brooklynne Mitchell-Arno, Oct 2025
